H. heilmannii is a Gram-negative bacterium resembling corkscrew (spiral shaped microorganisms) two to three times larger than H. pylori; 4 to 10 µm in length and 0.5 to 0.8 µm in diameter, has four to eight tight spirals. There are typically 8 to 12 tufts of bipolar flagella (10, 16, 17). It possesses the greatest number of hosts in mammals. It is present in the stomach of dogs, cats, leopards, rats, pigs and various species of primates. It rarely settles gastric mucosa of man. Haesebrouck et al. suggested using the name H. heilmannii sensu stricto to refer to the novel Helicobacter species and the name H. heilmannii sensu lato to refer to the whole group of non H. pylori Helicobacters (18). The prevalence of H. heilmannii infection in humans varies from 0.1% to 0.9% in patients presenting for upper gastrointestinal endoscopy, although it is reportedly higher in China (4%) and in Thailand where it is as high as 6% (14-16, 19, 20). H. heilmannii infection has an asymptomatic course, however it may lead to chronic gastritis, gastric and duodenal peptic ulcer and to other diseases in humans and animals. Diagnosis of H. heilmannii infection is made on the basis of bacterial morphology in direct microscopy of the specimen of the gastric mucosa and polymerase chain reaction (PCR). Attempts of cultures in vitro failed, but it is possible to sustain in vivo culture in laboratory animals (21).
The aim of the study was a clinical analysis of H. heilmannii infection in children, and the assessment of the incidence of H. heilmannii infection in children over a period of 18 years (1992-2010) according to age, sex, clinical symptoms and living environment.
Clinical analysis encompassed 22 children aged 4 to 18 years (11 girls and 11 boys) admitted and diagnosed in our clinic due to dyspeptic symptoms in whom H. heilmannii infection was diagnosed. The studied children were divided into two groups depending on the age. Into the first group nine children aged 4 to 13 years were included and into the second group children aged 14 to 18 years. In the studied children we analyzed a place of living (city, country), contact with domestic pets (dog, cat), clinical symptoms (epigastric pain, nausea, vomiting, heartburn) and the presence of concomitant diseases. In all children esophagogastroduodenoscopy was performed and specimens of mucosa from antrum were sampled for microbiology and histology studies. Erosive esophagitis, gastritis, duodenitis and ulcerative disease of the stomach and/or duodenum were taken into consideration in endoscopic diagnosis. The collected samples were studied by the mean of direct microscopy, microbiologic culture and in two children by PCR. Additionally in a part of the children urea test, IgG antibodies against H. pylori and study of H. pylori antigen in stool were performed. The specimens for direct microscopic examinations were stained using Gram stain method. Bacterial culture was conducted on medium containing Columbia agar with 7% of hemolyzed horse blood. The plates were incubated at 37°C in microaerophylic atmosphere (5% O2, 10%CO2, 85% N2) for 6 days. Anti H. pylori IgG antibodies were detected using ELISA test (enzyme linked immunoabsorbent assay) by Microgen-recom commercial kit. Concentration of antibodies above 24 u/ml was treated as positive. H. pylori antigen in stool specimens was detected by EIA method using the Amplified IDEIATMHpSTARTM test (DACO) according to the manufacturer's instruction.
In the period between 1992-2010 the number of 13,124 esophagogastroduodenoscopic studies were performed in children aged 4 to 18 years in the examination of 11,023 sampled specimens of gastric mucosa for Helicobacter infection. H. heilmannii infection was diagnosed in 22 children based on the examination of 11,023 samples, direct microscopic examination and culture (Fig. 1). The frequency of H. heilmannii infection was in 0.2% in direct microscopy of gastric mucosa specimens.
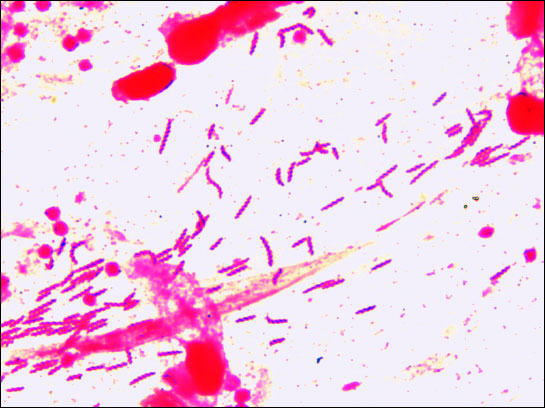 |
Fig. 1. Gram-stained direct smear of the antral mucosa infected with H. helmannii in examined patient (magnification 1000). |
Table 1 presents the data of the patients, clinical symptoms and endoscopic diagnosis. No sex difference in frequency of H. heilmannii infection was demonstrated (p>0,05). Most children lived in urban areas. 54.5% of the children had domestic contact with a dog and/or a cat. Most of the children belonged to the group aged 4 to 13 years. In all children chronic epigastric pain was observed, nausea in 45.4%, vomiting in 27.2% and heartburn in 13.6%. Nausea, vomiting and heartburn were observed more frequently in older children. Among endoscopic diagnoses dominated chronic nodular gastritis of antrum (77%), in 22.7% duodenitis. In single cases gastric or duodenal ulcer without H. pylori infection as well as erosive esophagitis were diagnosed. In three children (13.6%) endoscopic studies of the esophagus, stomach and duodenum did not reveal any changes but in microscopic study chronic inflammatory changes were observed. In children with endoscopic changes of histopatologic mucosa studies revealed chronic inflammatory process, which in half of the cases was active, infiltration of mononuclear cells or neutrophils were observed. In two children bronchial asthma was diagnosed, in three lactose intolerance and in four food allergy.
| Table 1. Patients, symptoms and results of esophagogastroduodenoscopy. |
 |
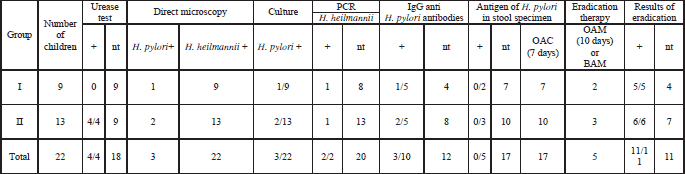
In Table 2 the results of Helicobacter diagnostic tests are presented. In three children mixed infection with H. pylori and H. heilmannii was diagnosed. In these children H. pylori infection was confirmed in direct microscopy of stomach biopsy specimen, positive culture and the presence of anti H. pylori IgG antibodies in serum. H. heilmannii infection was confirmed based on morphologic traits of Helicobacter in direct microscopy, negative culture for H. pylori and in some cases PCR, the lack of antibodies anti-H. pylori and the lack of H. pylori antigen in stool. All children with H. pylori and/or H. heilmannii infection were treated with proton pump inhibitor - omeprasole, amoxicillin and clarithromycin (OAC) for 7 days or with proton pump inhibitor, amoxicillin and metronidazole (OAM) for 10 days. Alternatively, instead of proton pump inhibitor - bismuth salts, amoxicillin and metronidazole (BAM) were administred. The result of eradication was examined after 8 weeks in 11 patients (50%). In all examined children eradication was successful. In 11 children the effect of eradication was not controlled.
| Table 2. The results of Helicobacter species identification. nt-not tested, O- proton pump inhibitor, A-amoxicillin, C-clarithromycin, M-metronidazole, B-bismuth subcitrate potassium. |
 |
H. heilmannii infection in children occurs relatively rarely. Based on earlier observations, the frequency of infections in children with dyspeptic symptoms was 0.5% (14). The present study on H. heilmannii infection based on a very large material demonstrated a rate of H. heilmannii infection to be 0.2% in direct microscopic examination of gastric mucosa specimens. Our results are similar to those in Europe: 0.3% in Bulgaria and 0.9% in Czech Republic. Also in other countries H. heilmannii infection is diagnosed rarely, for example 0.1% in Japan, with the exception of China - 4% and Thailand - 6.2% (15, 16, 20, 22). However, H. heilmannii infection is frequent in the animals. Interesting studies on Helicobacter spp infection in cats in our region have been conducted by Kubiak et al. (11). The authors studied species of Helicobacter in the stomach of 35 cats of European breed aged 1 to 10 years. Depending on the symptoms the cats were divided into two groups: the first one containing 10 cats without symptoms (control group) and the second group consisting 25 cats with dyspeptic symptoms (chronic nausea, fetor ex ore, lack of appetite, abdominal pain). Gastric biopsy samples taken from animals during endoscopy were analyzed by PCR. In the control group H. heilmannii infection was identified in seven cats (70%) including four cats with mixed infection with two species: H. heilmannii and H. felis. In the second group Helicobacter spp infection was found in 18 out of 25 animals (72%). H. heilmannii was present in five cats (27.8%), mixed infection with two species, H. heilmannii and H. felis or other species not identified by PCR, were observed in 13 cats (72.2%). The authors demonstrated a very frequent H. heilmannii infection in cats, both healthy and sick. Also in other studies frequent H. heilmannii infection in animals was demonstrated. Hwang et al. using PCR assay showed the presence of H. heilmannii in 85% and H. felis in 95% of cats (23). By contrast, in dogs H. heilmannii was observed in 76% of the cases while H. felis only in 4.8% (23). Moreover, the possibility of transmission of H. heilmannii infection from domestic animals to humans was proven. Gosciniak et al. described 14 years old girl living in a rural area in whom gastrointestinal endoscopy was conducted due to chronic abdominalgia (8). Chronic nodular gastritis and H. heilmannii infection has was diagnosed. Endoscopic study was also conducted in the dog and the cat with which the girl had had everyday contact. Endoscopic and histologic studies in the animals showed inflammation of the gastric mucosa with erosions and infection with the same species of H. heilmannii, which was proven by PCR, in the animals and the girl. The results of that study confirmed that H. heilmannii infection in the girl might have been of zoonotic origin (8). Other routes of infection also exist. Kato et al. documented H. heilmannii infection in 11-years-old boy three years after successful eradication of H. pylori and the healing of duodenal ulcer. The patient had had no contact with domestic animals, such as cats and dogs (24). In the material analyzed in our study 13 children (59.1%) with H. heilmannii infection had contact with a dog and/or a cat, however 9 children (40.1%) did not have any contact with domestic animals and the majority of them lived in urban area.
Diagnosis of H. heilmannii infection is based on morphology of biopsy specimens of the gastric mucosa, positive rapid urease test, and PCR. Culture, serum IgG anti-H. pylori antibodies test and stool test produced negative results. Our diagnostic studies allowed for diagnosis of mixed H. pylori and H. heilmannii infection in three children. Besides chronic modular gastritis, gastrointestinal examination of the patients infected with H. heilmannii revealed duodenitis (in five children) and gastric and duodenal ulcers (in two children). Histopathologic studies of the biopsy specimens from the prepyloric part of the stomach demonstrated a chronic inflammatory process of mild grade and in some of the children active inflammation of gastric mucosa. Previous experience in the treatment of H. heilmannii infection has indicated that H. heilmannii is sensitive to antibiotics used in the eradication of H. pylori (14, 15, 24). In our study on children H. heilmannii infection was successfully eradicated by the treatment with proton pump inhibitor, amoxicillin, clarithromycin or metronidazole.
In summary, H. heilmannii infection may be one of the causes of chronic gastritis and ulcerative disease in children. The diagnosis of H. heilmannii infection is generally made by the detection of its characteristic morphology in gastric biopsy specimens, since culture is extremely difficult and up till now has not been accomplished succesfully. H. heilmannii infection should be differentiated from H. pylori infection based on morphologic traits in direct microscopy, negative culture and absence of H. pylori antigen in stool.
Conflict of interests: None declared.
- Warren JR, Marshall BJ. Unidentified curved bacilli on gastric epithelium in active gastritis. Lancet 1983; 1: 1273-1275.
- Smet A, Flahou B, Mukhopadhya I, Ducatelle R, Pasmans F. The other Helicobacters. Helicobacter 2011; 16(Suppl. 1): 70-75.
- Stolte M, Wellens E, Bethke B, Ritter M, Edidt H. Helicobacter heilmannii (formerly Gastrosiorillum hominis) gastritis: an infection transmitted by animals? Scand J Gastroenterol 1994; 29: 1061-1064.
- Dent JC, McNulty CA, Ulf JC, Wilkinson SP, Gear MW. Spiral organisms in the gastric antrum [letter]. Lancet 1987; 2: 96.
- Dick E, Lee A, Wotson G, ORourke J. Use of the mouse for the isolation and investigation of stomach associated spiral-helical shaped bacteria trom man and other animals. J Med Microbiol 1989; 29: 55-62.
- Hanninen ML, Jalava K, Saari S, Happonen I, Wesermarcik F. Culture of "Gastrospirillum" from gastric biopsies of dogs. Eur J Clin Microbiol Infect Dis 1995; 14: 145-146.
- De Groote D, Ducatelle R, Haesebrouck F. Helicobacter of possible zoonotic origin: a review. Acta Gastroenterol Belg 2000; 63: 380-387.
- Gosciniak G, Skala J, Kubiak K, Iwanczak B, Biernat M, Grabinska J. The role of environmental Helicobacter heilmannii infection in etiopathogenasis of gastric diseases. Gastroenterol Pol 2006; 13: 320-323.
- Gosciniak G. Rodzaj Helicobacter - implikacje kliniczne i narastajacy problem opornosci Helicobacter pylori. Pediatr Wspolcz Gastroenterol Hepat Zyw Dziecka 2010; 12: 41-44.
- ORourke J, Grehan M, Lee A. Non-pylori Helicobacter species in human. Gut 2001; 49: 601-606.
- Kubiak K, Skala J, Nicpon J, Biernat M, Gosciniak G. The prevalence of Helicobacter species in the stomach of cats in Poland from Lower Silesia Area [abstract]. Helicobacter 2006; 11: 412.
- Yeomans ND, Kolt SD. Helicobacter heilmannii (formerly Gastrospirillum): association with pig and human gastric pathology. Gastronterology 1996; 111: 244-259.
- Yang H, Li X, Xu Z, Zhou D. Helicobacter heilmannii in a patient with gastric cancer. Dig Dis Sci 1995; 40: 1013-1014.
- Iwanczak F, Gosciniak G, Iwanczak B. Helicobacter heilmannii w patologii chorob zoladka i dwunastnicy u dzieci. Gastroenterol Pol 1998; 5: 229-233.
- Sykora J, Hejda V, Varvorovska J, Stozicky F, Siala K, Schwarz J. Helicobacter heilmannii gastroduodenal disease and clinical aspects in children with dyspeptic symptoms. Acta Pediatr 2004; 93: 707-709.
- Okiyama Y, Matsuzawa K, Hidaka E, Sano K, Akamatsu T, Ota H. Helicobacter heilmannii infection: clinical, endoscopic and histopathological features in Japanese patients. Pathol Int 2005; 55: 398-404.
- Andersen LP. New Helicobacter species in humans. Dig Dis 2001; 19: 112-115.
- Haesebrouck F, Pasmans F, Flahou B, Smet A, Vandamme P, Ducatelle R. Non Helicobacter pylori Helicobacter species in the human gastric mucosa: a proposal to introduce the names H. heilmannii sensu lato and sensu stricte. Helicobacter 2011; 16: 339-340.
- Heilmann KL, Borchard F. Gastritis due to spiral shaped bacteria other than Helicobacter pylori: clinical, histological, and ultrastructural findings. Gut 1991; 32: 137-140.
- Yali Z, Yamada N, Wen M, Matsuhisa T, Miki M. Gastrosprillum hominis and Helicobacter pylori infection in Thai individuals: comparison of histoapthological changes of gastric mucosa. Pathol Int 1998; 8: 507-511.
- Solnick JV, Schumer DB. Emergence of diverse Helicobacter species in the pathogenesis of gastric and enterohepatic diseases. Clin Microbiol Rev 2001; 14: 59-97.
- Boyanowa L, Koumanova R, Lazarova E, Jelev C. Helicobacter pylori and Helicobacter heilmannii in children. A Bulgarien study. Diagn Microbiol Infect Dis 2003; 46: 249-252.
- Hwang CY, Han R, Youn HY. Prevalence and clinical characterization of gastric Helicobacter species infection of dogs and cats in Korea. J Vet Sci 2002; 3: 123-133.
- Kato S, Ozawa K, Sekine H, et al. Helicobacter heilmannii infection in a child after successful eradication of Helicobacter pylori: case report and review of literature. J Gastroenterol 2005; 40: 94-97.