DIVERGING OXIDATIVE DAMAGE AND HEAT SHOCK PROTEIN 72 RESPONSES TO ENDURANCE TRAINING AND CHRONIC TESTOSTERONE PROPIONATE TREATMENT IN THREE STRIATED MUSCLE TYPES OF ADOLESCENT MALE RATS
INTRODUCTION
Heat shock proteins (Hsp) play a critical role in maintaining cellular homeostasis. The diverse identified families of Hsp act as molecular chaperones or cochaperones and stress, sensors and bestow direct protection or rescue to the endangered or injured cells (1, 2). Exposure to various stressful conditions, e.g. hypoxia, hyperthermia, or oxidative or mechanical stress typically enhances expression of a variety of Hsp in diverse tissues and organs and may harden them to repeated environmental challenges (3-6). In striated muscles, the most common stressor is physical effort that primarily enhances the expression of the so-called inducible 72 kDa Hsp (Hsp72, also called Hsp70) (7-10). Increased Hsp72 expression may assure complementary protection against exercise-induced increase in oxidative stress (11). Diverse striated muscle types show differing Hsp72 responses to both heat stress and exercise training of various types and intensities (12, 13).
Testosterone and other anabolic-androgenic steroids (AAS) have been used for many years by athletes with the aim to increase muscle volume and strength (14, 15) or just to speed up bodybuilding (16). These effects correlate positively with AAS dosage over a wide range (17), which link promotes AAS abuse at high doses. AAS were also reported to improve submaximal running endurance in male rats when applied concurrently with endurance training (18, 19). Intriguingly, this was despite no difference in maximum oxygen consumption, running economy and red blood cell parameters between the trained rats given AAS and those given the respective vehicle (19). However, no positive effect of AAS on endurance performance was obtained in a human setting (20) and some other rat studies (21).
AAS abuse becomes increasingly popular in adolescents, mainly boys, who often combine it with physical training (15, 22, 23). However, the possible harmful health effects of this combination in the youth, in whom the related oxidative stress and endocrine phenomena may interfere with ongoing somatic growth and the still-developing pattern of sex steroids' milieu (24-26), were given little attention. This was despite both the observation that junior and senior subjects differ in their responses to oxidative stress in at least some types of endurance training (26, 27) and the decreasing age at which teenagers engage in competitive sports. Illicit testosterone supplementation is mostly used in combination with strength training or bodybuilding. Unfortunately, there is no rat model of strength training involving muscle masses comparable (in relative terms) to those engaged in such training in humans. However, endurance exercise is considered an important complement of strength training in some sport disciplines and is also recommended in modern bodybuilding programs (see http://www.bodybuilding.com/fun/wiggy1.htm or http://www.teenbodybuilding.com/locke17.htm). The main goal of this addition is to enhance oxidative capacity and promote body-sculpting by enhancing lipid metabolism. As the data on the effects of combining this exercise type with AAS abuse are scanty, we decided to test this combination in our studies.
We previously modeled (28) the effects of chronic testosterone treatment and endurance training on enzymatic and low-molecular weight nonenzymatic components of anti-oxidative stress myocardial defenses. The present study was aimed at typifying the effects of these factors on yet another line of cellular anti-stress defenses that relies on the expression of Hsp72. Hence, we compared the effects of these treatments on lipid peroxidation, content of the inducible Hsp72 and the expression of androgen receptor (AR) in selected striated muscles of adolescent male rats. While all these muscles are androgen targets well-known for their adaptation to exercise, they greatly differ in metabolic properties. Our choice was based on the fact that the heart left ventricle (LV) is of key importance for proper supply of blood for the working muscles during endurance exercise, and the soleus (Sol) and the extensor digitorum longus (EDL) represent, respectively, skeletal muscles that primarily use oxidative (aerobic) metabolism and those that rely predominantly on glycolytic (anaerobic) metabolism.
MATERIALS AND METHODS
Animals and experimental design
Five-week old male Wistar rats of 100–120 g initial body weight (b.w.) from the stock of the Mossakowski Medical Research Centre in Warsaw were used for the study. The rats were kept on a 12/12-h light/dark cycle (lights on at 7 a.m.) in a temperature (22–24°C) and humidity (45–65%) controlled room, with free access to standard rat chow and purified tap water. Before the start of training, all rats were tested on a motorized rodent treadmill to habituate them to this setting and eliminate rats reluctant to run (28). The preselected rats were randomized into six groups: 1) untrained (sedentary, UTr) controls, 2) untrained rats given 8 mg/kg b.w. of testosterone propionate (TP; Jelfa, Poland) weekly (Utr + TP8), 3) untrained rats given 80 mg/kg b.w. of TP weekly (Utr + TP80), 4) TP-untreated trained rats (Tr), 5) trained rats given 8 mg/kg b.w. of TP weekly (Tr + TP8), and 6) trained rats given 80 mg/kg b.w. of TP weekly (Tr + TP80). The basis for the choice of TP doses was given earlier (28). TP was diluted with sesame oil as needed to ensure equal injection volume for both dosages, and was given intramuscularly once a week, 1 hour after running session; TP-untreated rats received the same volume of the oil by this schedule. The rats scheduled for training were exercised on the treadmill (at 0° inclination) 5 days/week for 6 weeks, starting with a 40 min session at 16 m/min. Exercise duration and intensity were gradually increased over the first 4 weeks and then were kept steady at 60 min daily and 28 m/min. This training scheme has been shown by biochemical, morphological and performance measures to be very effective in terms of endurance improvement (29-31). Core body temperature (Tc) was measured in the trained rats just before and instantly after the last training session, using a flex-tip digital thermometer inserted 1.5–2 cm deep into the rectum; simultaneously, resting Tc was measured in their untrained counterparts. Two days after the session, all rats were anesthetized by intraperitoneal sodium pentobarbital injection (Vetbutal, Polfa, Poland; 50 mg/kg b.w.) and decapitated. Trunk blood was collected and processed for serum that was stored at –80°C for testosterone assay. Chests were opened instantly and the rats were perfused (for 3 min, at 25 ml/min) through the LV with glucose-supplemented (10 mM) O2/CO2 (95%/5%, v/v) mixture-saturated Krebs-Henseleit buffer. Then the LV, the Sol and the EDL from both hind limbs were dissected, snap-frozen in liquid nitrogen and stored at –80°C for Hsp72, AR gene expression and thiobarbituric acid-reactive substances' (TBARS) assays. The study protocol has been accepted by the First Local Ethics Committee for Animal Experimentation, Warsaw, Poland (Certificate of approval No. 251).
Biochemical assays
Tissue samples meant for Hsp72 assay by Western blotting were homogenized with an Ultra-Turrax homogenizer (IKA-Werke, Staufen, Germany) in 3 volumes of an ice-cold 10 mM Tris-HCl buffer (pH 7.5) containing 0.1 mM phenylmethanesulfonyl fluoride and 250 mM sucrose, and then were spun at 100 000 × g for 1 hour. The obtained cytosols were assessed for protein content and next were diluted 1:3 with sample buffer (200 mM Tris-HCl, 10% SDS, 30% glycerol, 0.2% bromophenol blue; pH 6.8) and heated for 4 min in a water bath (90°C). The denatured cytosols (25 µg protein/lane) were mixed with 10–180 kDa Prestained Protein Ladder (Fermentas, Cambridge, UK; 3 µl/lane) and loaded onto 10% acrylamide gel overlaid with 4% stacking gel. Electrophoresis was run at 15 V/cm for 90 min, using a 2 mM Tris, 192 mM glycine, 0.1% SDS buffer pH 8.3. Resolved proteins were transferred onto nitrocellulose membranes with the Mini-Protean 3 Cell/Mini Trans-Blot Module/PowerPac system (BioRad, Hercules, CA), at 4°C. Next, the membranes were blocked with 5% (w/v) solution of nonfat milk powder in Tris-buffered saline (TBS: Tris-HCl 1M, NaCl 2M, pH 7.4), rinsed for 1 min in TBS supplemented with 0.05% (v/v) Tween-20 (TBST), incubated overnight at 4°C with the anti-Hsp72 monoclonal antibody SPA-810 (Stressgen, Ann Arbor, MI) diluted 1:1000 with the milk-supplemented TBS, rinsed again (3 × 10 min) with TBST and incubated for 1 hour with a horseradish peroxidase-conjugated anti-mouse IgG (Vectastain ABC kit, Vector Laboratories Inc., Burlingame, CA) diluted 1:1000 with the milk-supplemented TBS. Then the blots were washed in TBST (3 × 10 min), developed in 3% H2O2/0.2 mM diaminobenzidine solution in 0.5 mM Tris-HCl buffer, pH 7.4, and quantified on a NucleoVision densitometer and the GelExpert 4.0 software (Nucleo-Tech Corp., San Mateo, CA). After the quantitation, the membranes were subjected to a standard stripping process, blocked again with the milk-supplemented TBS, and incubated overnight at 4°C with rabbit polyclonal anti-GAPDH antibody G9545 (Sigma) diluted 1:10,000 with TBST supplemented with 0.1% (w/v) BSA. Next, the membranes were incubated for 1 hour at room temperature with goat anti-rabbit IgG-horseradish peroxidase conjugate (diluted 1:8000 with TBST supplemented with 5% (w/v) nonfat milk powder). GAPDH-containing immune complexes were visualized using an ECL system and quantified with the NucleoVision densitometer and the Gel Expert 4.0 software.
AR gene expression was assessed by real-time reverse transcriptase-polymerase chain reaction (RT-PCR) method. Total RNA was extracted from tissue samples with TRI-reagent (Sigma, St. Louis, MO, USA); contaminating DNA was digested with DNase I (Sigma). Reverse transcription of 2 µg of the DNA-free total RNA was run in a total volume of 20 µl using high capacity cDNA reverse transcription kit (Applied Biosystems, Foster City, CA, USA), in one cycle (25°C for 5 min, 42°C for 45 min, and 85°C for 5 min, with subsequent cooling to 4°C). Multiplex RT-PCR was run in triplicates on a model 7500 real-time PCR system (Applied Biosystems) using rat AR-specific primers/probe set Rn00560747 m1, rat β-actin-specific ACTB endogenous control primers/probe set and TaqMan Universal PCR Master Mix (Applied Biosystems all), and 5 µl of the reverse transcription product, in a total volume of 20 µl. PCR conditions were as follows: 10 min denaturation at 95°C followed by 50 cycles of 95°C for 15 s and 60°C for 1 min. AR gene expression level was calculated using the standard curve method and normalized to actin-β expression.
TBARS were measured by the method of Ohkawa et al. (32), with minor modifications (28). Serum total testosterone (TT) level was assayed with an RIA kit (DSL-4100; Diagnostic System Laboratories, Webster, TX, USA), and tissue protein content was determined by the 2,2'-bicinchoninic acid method using the Sigma protein assay kit BCA1.
Statistical analysis
Data are shown as the mean ± S.D. All data were first tested for homogeneity of variance by the Levene test. TBARS and Hsp72 contents' data were acquired from differing rat numbers for the various muscles studied; hence they were then analyzed by a 3-way ANOVA with TP dose (0, 8, or 80 mg/kg weekly), muscle type (LV, Sol, and EDL) and training status (Tr or UTr) as the main factors, followed by the Tukey test for unequal sample sizes. AR expression data, which were acquired from all the three studied muscle types in each rat, were analyzed by repeated measures 3-way ANOVA with muscle type as the repeated measures factor, followed by the Tukey test for equal sample sizes. Since the 3-way ANOVA of AR data gave a significant muscle type × training × TP dose interaction effect, the significance of between-group differences was tested for each muscle type separately using a 2-way ANOVA followed by the Tukey test. TT levels and exercise-related changes in Tc were analyzed by a 2-way ANOVA with training status and TP dose as the main factors, followed by the Tukey test and the Student-Newman-Keuls test, respectively. Relationships between different variables were tested by Spearman's rank correlation test. In all cases, a p<0.05 was considered significant. All the analyses were performed using the Statistica 7.1 software (StatSoft, Tulsa, OK, USA).
RESULTS
Effects of testosterone propionate treatment and endurance training on blood serum total testosterone levels and weight of the selected striated muscles
Two-way ANOVA yielded significant effects of TP dose (F2,30=582.7, p<10-3) and training × TP dose interaction (F2,30=5.68, p=0.008), but not of the training (F1,30=2.67, p=0.11), on the TT levels. Blood serum TT levels were significantly and markedly higher in the TP-untreated endurance-trained rats than in their sedentary counterparts, while there was no difference in TT levels between trained and untrained rats given identical TP treatment (Fig. 1). Two-way ANOVA showed no effect of experimental manipulations on LV weight, whereas it yielded significant effect of the training on Sol weight and of the TP treatment on EDL weight. However, these main factor effects did not translate into statistically significant differences between the respective individual study groups (Table 1).
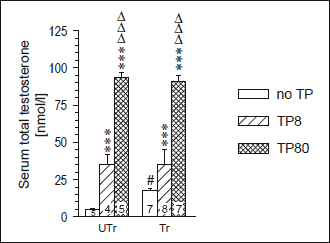 |
Fig. 1. Blood serum total testosterone level in sedentary (UTr) and endurance-trained (Tr) adolescent male Wistar rats given weekly intramuscular injections of testosterone propionate (TP8 8 mg/kg, TP80 80 mg/kg; 6 doses). The data were taken, under the Creative Commons Attribution limits, from an earlier work of the authors (28). For TP treatment, training and blood collection details see Materials and methods. *** p<0.001 vs. the respective TP-untreated group, # p<0.05 vs. the respective UTr group, ΔΔΔp<0.001 vs. the respective low-dose TP-treated group; the Tukey test. Numbers of rats are shown inside the respective bars. |
Effects of testosterone propionate and endurance training on lipid peroxidation
Three-way ANOVA showed significant effects of muscle type, training and TP dose on TBARS content (F2,94=232.6, p<10-3; F1,94=48.0, p<10-3; and F2,94=163.6, p<10-3; respectively), and muscle type × training and muscle type × TP dose interactions (F2,94=3.60, p=0.031, and F4,94=22.6, p<10-3, respectively), but no significant training × TP dose (p=0.55) or training × TP dose × muscle type (p=0.10) interaction. Training alone significantly elevated TBARS only in the Sol, while TP treatment alone (at either dose used) showed much stronger effect, more than doubling TBARS content in the LV and Sol, and almost doubling it in the EDL. Increasing TP dose above 8 mg/kg b.w. did not translate into further significant increases in TBARS level in any muscle studied, either in untrained or in trained rats. There was no difference in TBARS level between the LV and Sol under any experimental condition, whereas the EDL showed significantly lower level of lipid peroxidation than the other studied muscles under all these conditions except in sedentary TP-untreated rats (Fig. 2).
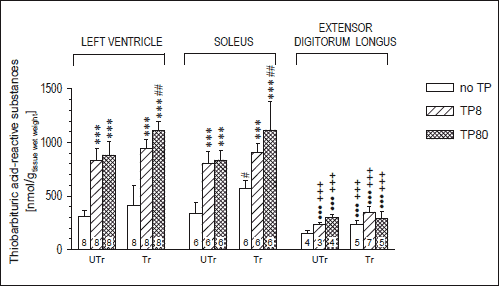 |
Fig. 2. TBARS content in various muscles of sedentary (UTr) and endurance-trained (Tr) adolescent male Wistar rats given weekly intramuscular injections of testosterone propionate (TP8 8 mg/kg, TP80 80 mg/kg; 6 doses). For TP treatment and training details see Materials and methods. *** p<0.001 vs. the respective TP-untreated group; #p<0.05, ##p<0.01, ●●●p<0.001 vs. the respective Sol value; +++p<0.001 vs. the respective LV value. Numbers of rats are shown inside the respective bars. The LV data were taken, under the Creative Commons Attribution limits, from an earlier work of the authors (28). |
Effects of testosterone propionate treatment and endurance training on heat shock protein 72 content
Three-way ANOVA showed significant effects of muscle type, endurance training and TP dose on Hsp72 content (F2,51=242.9, p<10-3; F1,51=3363.6, p<10-3; and F2,51=23.1, p<10-3; respectively), significant muscle type × training and TP dose × training interaction effects (F2,51=177.2, p<10-3, and F2,51=34.4, p<10-3), but not muscle type × TP dose (p=0.48) or muscle type × TP dose × training (p=0.22) interaction effect. Under basal conditions, Hsp72 level was but negligibly higher in the LV and Sol than in the EDL. Endurance training alone elevated Hsp72 relatively more in the LV and Sol than in the EDL (+528, +506 and +302%, respectively). TP treatment dose-dependently up-regulated Hsp72 in all studied muscles in sedentary rats, but not in their endurance-trained counterparts. Contrariwise, the lower TP dose slightly, but significantly, counteracted the effect of the training in the LV and EDL (Fig. 3).
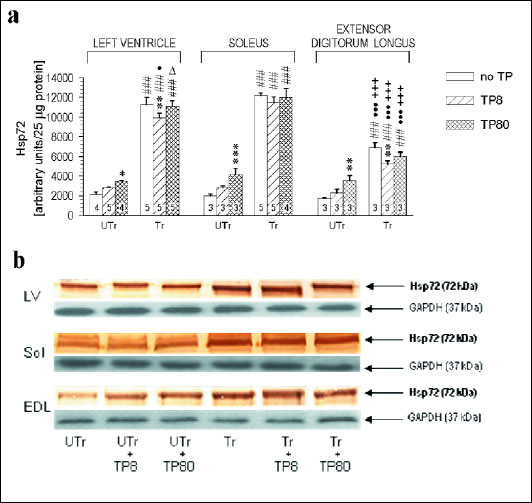 |
Fig. 3. (a) Hsp72 contents in various muscles of sedentary (UTr) and endurance-trained (Tr) adolescent male Wistar rats given weekly intramuscular testosterone propionate injections (TP8 8 mg/kg, TP80 80 mg/kg; 6 doses). For TP treatment and training details see Materials and methods. * p<0.05, ** p<0.01, *** p<0.001 vs. the respective TP-untreated group; ###p<0.001 vs. the respective UTr group; Δp<0.05 vs. the respective low-dose TP-treated group; ●p<0.05, ●●●p<0.001 vs. the respective Sol value; +++p<0.001 vs. the respective LV value; the Tukey test. (b) Representative Western blots showing Hsp72 and GAPDH expression in the heart left ventricle (LV), in the soleus (Sol) and in the extensor digitorum longus (EDL) of TP-treated and -untreated sedentary and endurance-trained rats. Numbers of rats are shown inside the respective bars. |
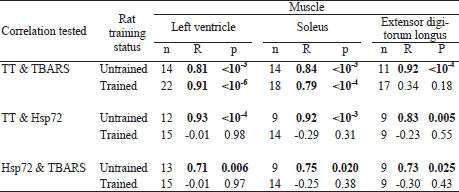
To verify the possibility that the changes in TBARS and Hsp72 contents may be related to experimentally induced changes in TT or mutually related, correlations between these variables were tested by the Spearman rank correlation test (Table 2).
Effects of testosterone propionate and endurance training on AR mRNA level
Repeated measures 3-way ANOVA yielded significant effects of muscle type and training (F2,36=746.2, p<10-3, and F2,36=283.4, p<10-3; respectively), but not of TP dose (F1,36=2.62, p=0.12), and significant TP dose × training, muscle type × training, muscle type × TP dose and muscle type × TP dose × training interaction effects (F2,36=166.8, p<10-3, F2,36=9.47, p<10-3, F4,36=64.0, p<10-3 and F4,36=34.0, p<10-3, respectively). Due to the triple interaction effect, the data were next analyzed for each muscle separately. Two-way ANOVA gave significant TP dose and TP dose × training interaction effects in the LV (F2,18=5.32, p=0.015, and F2,18=9.12, p=0.002, respectively), Sol (F2,18=122.3, p<10-3, and F2,18=50.3, p<10-3, respectively) and EDL (F2,18=140.6, p<10-3, and F2,18=99.1, p<10-3, respectively), and significant training effect in the EDL only (F1,18=12.4, p=0.0025). AR mRNA level was markedly lower in the LV than in the Sol and EDL. Training alone significantly elevated it in all these muscles (+82%, +54% and +164%, respectively). In sedentary rats, low- and high-dose TP treatment, respectively, significantly increased and reduced AR mRNA in the Sol and EDL (+50% and 22%, and +65% and 53%, respectively), but showed no sizeable effect in the LV (+23% and 2%, respectively, pł0.82). In trained rats, both the low- and high-dose TP treatment significantly reduced AR gene expression in the Sol (26% and 75%, respectively), EDL ( 64% and 89%, respectively) and LV ( 48% and 45%, respectively); (Fig. 4).
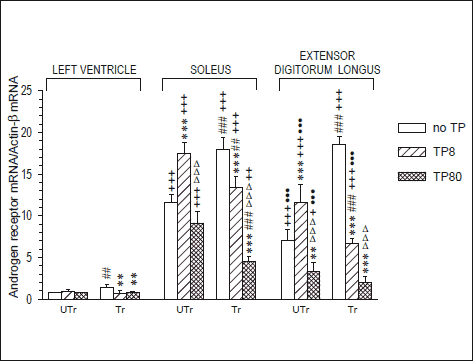 |
Fig. 4. Androgen receptor gene expression in various muscles of sedentary (UTr) and endurance-trained (Tr) young male Wistar rats given weekly intramuscular testosterone propionate injections (TP8 8 mg/kg, TP80 80 mg/kg; 6 doses); N=4 for each group. For TP treatment and training details see Materials and methods. ** p<0.01, *** p<0.001 vs. the respective TP-untreated group; ##p<0.01, ###p<0.001 vs. the respective UTr group, ΔΔΔp<0.001 vs. the respective low-dose TP-treated group; ●●p<0.01, ●●●p<0.001 vs. the respective Sol value; +p<0.05, ++p<0.01, +++p<0.001 vs. the respective LV value; the Tukey test. Please note that the differences between the various muscles were tested for significance by the post-hoc test for 3-way ANOVA, whereas the between-group differences were tested for significance by the post-hoc test for 2-way ANOVA that was run for each muscle separately. |
Effects of testosterone propionate treatment and endurance training on core body temperature
There was no difference in resting Tc between study groups (one-way ANOVA: F5,42=1.32, p=0.28). The final training session significantly elevated Tc in all endurance-trained rat groups. TP-treated rats showed significantly smaller rises in Tc (ΔTc) than their TP-untreated counterparts, and the rats treated with high TP dose showed larger than those given the lower dose (Table 3). Spearman rank correlation test yielded a moderate negative correlation between serum TT and ΔTc: R= –0.44, p=0.042 (n=22).

DISCUSSION
The data on the effects of physical training on antioxidant defenses and oxidative stress are numerous, but inconsistent (33, 34). This is probably due to disparities among various studies in training type and subjects' health, sex, age and hormonal/metabolic status. Notably, adolescent males experience a dramatic testosterone-related expansion of muscle size (35, 36). Our choice of endurance training was due to the fact that it plays a key role in many sports and gets more and more consideration as a health-promoting factor. The main findings of this study are as follows: 1) chronic TP treatment alone enhanced lipid peroxidation markedly more in the LV and Sol than in the EDL; 2) the training alone markedly elevated Hsp72 in all these muscles; 3) the TP treatment alone dose-dependently but slightly upregulated Hsp72 in sedentary, but not in endurance-trained rats; 4) the training alone enhanced AR gene expression in all studied muscles; 5) low-dose TP treatment alone enhanced AR gene expression in the Sol and EDL, whereas high-dose TP treatment alone suppressed it in the EDL, but not in the other muscles; 6) in the trained rats, chronic TP treatment dose-dependently suppressed AR gene expression in all these muscles.
Hsp72 is constitutively expressed at basal levels in all cell types as one of the so-called housekeeping proteins (37). Besides heat stress, the most important factor that enhances its production is oxidative stress (38). In skeletal muscles, Hsp72 upregulation is a typical response to contraction-linked energy and milieu challenges (13). The endurance training-related increases in Hsp72 levels found in the present study are in line with earlier reports (6, 11, 39-42) with some divergence mostly attributable to differences in the type, intensity and duration of the exercise or training.
The present data indicate that the key factor in Hsp72 upregulation in endurance-trained adolescent rats was the exercise-related increase in Tc. This is because: 1) TP treatment dose-dependently enhanced oxidative stress (as evidenced by increased TBARS content) in most studied muscles, but it counteracted the training-related upregulation of HspSP72 in these rats, as indicated by significant TP dose × training interaction effect (see also Fig. 3); and 2) the effect of TP treatment on Hsp72 contents roughly paralleled its effect on the exercise-related Tc increase (Table 3). Interestingly, some authors (42) reported a sizeable increase in heart Hsp72 level in adolescent male rats only after a relatively long (>12 weeks) period of endurance training, which suggests that the exercise-related increase in Tc may not be a dominant factor in Hsp72 upregulation in this organ. However, TP treatment-related increases in Hsp72 content were also found in all tested muscles of sedentary rats, with no change in resting Tc. This might suggest 'direct' enhancement of Hsp72 expression by testosterone, but this guess lacks a rationale or an experimental support. More likely, this effect was due to the TP treatment-related increase in oxidative stress. No added increase in Hsp72 content by this mechanism was apparent in endurance-trained rats despite significant elevations in TBARS. This could be due to a 'ceiling' effect of the training on Hsp72 content and/or masking of the effect of TP by the much stronger effect of the training. This possibility finds support in the training-related abolition of the correlation between serum TT and tissue Hsp72 levels (Table 2). Still, while the low TP dosage partly counteracted the effect of training on Hsp72 levels, the high-dose TP treatment showed no such action. This suggests a dual effect of TP treatment on Hsp72 expression: 1) an upregulation related to enhanced oxidative stress, and 2) an opposite effect consequent on the TP treatment-related suppression of exercise-related increase in Tc, of which the former apparently played a major role. The fact that the decrease was smaller after the high-dose TP treatment might be the consequence of markedly reduced AR gene expression. This explanation implies also that the effects of TP treatment on lipid peroxidation and Hsp72 expression are mediated by distinct mechanisms, as muscle TBARS contents showed a general tendency for elevation with increased TP dose.
The ΔTc-lowering effect of testosterone was also reported in castrated male Japanese quails (43) and in C57B1/6 mice (44). Altogether, these observations imply that this effect is only at play when Tc is elevated for some reason. Another explanation for the negative impact of high serum TT level on Hsp72 expression could be an AR-mediated suppression of hsp72 gene expression, which effect was found in rat hearts and cultured cardiomyocytes after hyperthermia followed by testosterone treatment (45). Interestingly, the training we used did not affect by itself the exercise-related rise in Tc while it markedly elevated circulating TT. We did not assess the effect of exercise on Tc in untrained rats in this study as this would bias baseline TBARS levels. Yet the exercise-related Tc in the UTr group rats (Table 3) did not significantly differ (P=0.10, two-tailed t-test) from that found in a group of age-matched naive male Wistar rats after a single session of treadmill running at the maximum duration and intensity used in this study (n=10, ΔTc ± S.D: 3.4±0.3°C; unpublished data from our lab).
A common feature of various heat shock factor-activating conditions, which activation is a crucial intermediate step in Hsp72 upregulation, is increased generation of reactive oxygen species (ROS) (46, 47) resulting, inter alia, in enhanced lipid peroxidation and TBARS formation. In this study, the major factor in the damage could be high serum TT level. Significant TP treatment-related elevation of TBARS in the Sol and LV, but not in the EDL, is in an apparent conflict with the known higher capacity of anaerobic muscle fibers for ROS production (48). One reason for the disparity between TP effects on TBARS level in the various muscles might be differences in muscle sensitivity to androgens. There is evidence that endurance training upregulates AR and thus may elevate the sensitivity in rat LV myocytes (49) and in the Sol, but not in the EDL (50). Other data contradict this possibility, see (51), and so do the results of the present study. First, the training alone enhanced AR gene expression in the EDL as well. Second, it was not the AR mRNA-rich EDL, but the AR mRNA-poor LV that showed marked TP treatment-related rises in TBARS. This suggests that AR is not a major determinant of striated muscle reactivity to androgens. Notably, AAS can act via nongenomic pathways that include both AR- and non-AR-mediated mechanisms (52-54). There is also evidence that not all muscle cells within a given striated muscle express AR, see (49) and the respective references therein. Hence, changes in AR expression may not be a reliable predictor of changes in muscle responsiveness to testosterone. There is also evidence that various skeletal muscles differ in the pathways that mediate a given response to androgens (54). However, the differences in TP effect on TBARS level in the various muscles might also be related to higher availability of unsaturated fatty acids that are the major substrate for TBARS formation (55) in aerobic compared with anaerobic muscles (56). Interestingly, as evidenced by TBARs titers, there was no support for a beneficial effect of endurance training on muscular lipid peroxidation in our study, whereas a circumstantial evidence for this effect was found in healthy young adult men (57). This may suggest an elevated risk of exercise-related oxidative damage in adolescence.
Testosterone can shift prooxidant/antioxidant balance toward the prooxidant state and increase lipid peroxidation in various AR-positive cells, including myocytes (58-60), but this effect may not be AR-mediated (61). It seems also doubtful to result from testosterone aromatization and the consequent stimulation of ROS formation, as it can be induced with the non-aromatizable androgen 5a-dihydrotestosterone (62) and the TP treatment caused no major elevation of circulating estradiol (28). On the other hand, the synthetic nonaromatizable AAS stanozolol that compared to testosterone shows low affinity for AR (63) and very low androgenic/anabolic ratio (15) was found to cause no increase in Sol and EDL TBARS contents when given at a dose of 2 mg/kg b.w., 5 days/week, for 8 weeks, to adolescent male rats (21). Whereas chronic TP treatment elevated TBARS considerably more in the LV and Sol than in the EDL, there were only minor differences in relative magnitudes of this effect for the two TP doses used. This may be due to the fact that the blood TT levels attained with either TP dose exceeded by an order of magnitude the rat AR dissociation constant (64), suggesting near-saturation of the receptor. More importantly, the effects of TP treatment on AR gene expression showed different dose-effect relationship in sedentary compared to endurance-trained rats, whereas there was no such difference in the respective changes in TBARS contents. This suggests that the effect of TP treatment on lipid peroxidation could be mediated by nongenomic pathways.
Our finding of positive association between tissue TBARS and serum TT levels confirms pro-oxidant action of elevated testosterone. AR is present in both cardiomyocytes (65) and in slow-twitch (aerobic) and fast-twitch (anaerobic) muscle fibers in rodents (54). Testosterone stimulates hormone-sensitive lipase and enhances lipolysis in rat cardiomyocytes (66) and likely also in the Sol, but less so in the EDL (67). The possible rise in fatty acids availability for ATP synthesis suggests increased oxygen use and enhanced ROS generation and oxidative stress. It is thus reasonable to assume that the latter effect would be more evident in aerobic (e.g. Sol) than in anaerobic (e.g. EDL) muscles (68).
The endurance training-related increases in muscle TBARS contents were relatively low in this study compared with studies on exercise-related oxidative damage in skeletal muscles of young adult rats (27, 69). This disparity may be due, at least partly, to differences in exercise/training protocols. In rats treated with the lower TP dose (8 mg/kgBW), the effect of endurance training might have been further obscured by the apparently near-maximal lipid peroxidation damage for a given muscle type. On the other hand, the significant TBARS-elevating effect of the increase in TP dosage, which was apparent in the LV and Sol, could be related to the aforementioned effect of testosterone on lipolysis and the resulting increased supply of substrate for lipid peroxidation. This possibility is supported by little impact of endurance training on the correlation between TBARS and serum TT in the LV and Sol that remained close to that found in sedentary rats (Table 2). The latter was not true for the EDL that has relatively small supply of unsaturated fatty acids (56), and thus endurance training alone might be sufficient for maximal enhancement of ROS formation and TBARS production in this muscle.
The question arises whether and which of the effects observed in our study could be related to the influence of endurance training on maturational development of the constituent fiber types in the various studied muscles, as such influence was observed in adolescent male mice (70) and the muscles considered in our study differed considerably in fiber type composition. Answering this question warrants an additional investigation.
In summary, the results of this study indicate that chronic treatment with suprapharmacological TP doses may exert, due to upregulation of Hsp72 expression, a protective effect on muscle cell proteins in sedentary, but not in endurance-trained adolescent male rats. The up-regulation of Hsp72 in sedentary male adolescent rats subject to high-dose TP treatment is stimulated by androgen-induced oxidative stress, but it seems unlikely that this effect results from AR-mediated direct genomic action of testosterone. Exercise-related heat stress, and not oxidative stress, is the main factor responsible for up-regulation of Hsp72 expression in the myocardium and skeletal muscles of endurance-trained and TP-treated adolescent male rats.
Acknowledgments: This study was supported by the grant #6P05A-086-21 from the State Committee for Scientific Research (Warsaw, Poland) and by statutory funds from the Jerzy Kukuczka Academy of Physical Education, Katowice, Poland.
Conflict of interests: None declared.
REFERENCES
- Siu PM, Bryner RW, Martyn JK, Always SE. Apoptotic adaptations from exercise training in skeletal and cardiac muscles. FASEB J 2004; 18: 1150-1152.
- Welch WJ. Mammalian stress response: cell physiology, structure/function of stress proteins, and implications for medicine and disease. Physiol Rev 1992; 72: 1063-1081.
- Locke M. The cellular stress response to exercise: role of stress proteins. Exerc Sport Sci Rev 1997; 25: 105-136.
- Snoeckx LH, Cornelussen RN, van Nieuwenhoven FA, Reneman RS, van der Vusse GJ. Heat shock proteins and cardiovascular pathophysiology. Physiol Rev 2001; 81: 1461-1497.
- Ruell PA, Hoffman KM, Chow CM, Thompson MW. Effect of temperature and duration of hyperthermia on HSP72 induction in rat tissues. Mol Cell Biochem 2004; 267: 187-194.
- Tarricone E, Scapin C, Vitadello M, et al. Cellular distribution of Hsp70 expression in rat skeletal muscles. Effects of moderate exercise training and chronic hypoxia. Cell Stress Chaperones 2008; 13: 483-495.
- Harris MB, Starnes JW. Effects of body temperature during exercise training on myocardial adaptations. Am J Physiol Heart Circ Physiol 2001; 280: H2271-H2280.
- Atalay M, Oksala NK, Laaksonen DE, et al. Exercise training modulates heat shock protein response in diabetic rats. J Appl Physiol 2004; 97: 605-611.
- Whitham M, Fortes MB. Heat shock protein 72: release and biological significance during exercise. Front Biosci 2008; 13: 1328-1339.
- Ogura Y, Naito H, Akin S, et al. Elevation of body temperature is an essential factor for exercise-increased extracellular heat shock protein 72 level in rat plasma. Am J Physiol Regul Integr Comp Physiol 2008; 294: R1600-R1607.
- Smolka MB, Zoppi CC, Alves AA, et al. HSP72 as a complementary protection against oxidative stress induced by exercise in the soleus muscle of rats. Am J Physiol Regul Integr Comp Physiol 2000; 279: R1539-R1545.
- Oishi Y, Taniguchi K, Matsumoto H, Ishihara A, Ohira Y, Roy R. Differential responses of HSPs to heat stress in slow and fast regions of rat gastrocnemius muscle. Muscle Nerve 2003; 28: 587-594.
- Liu Y, Gampert L, Nething K, Steinacker JM. Response and function of skeletal muscle heat shock protein 70. Front Biosci 2006; 11: 2802-2827.
- Wilson JD. Androgen abuse by athletes. Endocr Rev 1988; 9: 181-199.
- Hoffman JR, Kraemer WJ, Bhasin S, et al. Position stand on androgen and human growth hormone use. J Strength Cond Res 2009; 23(Suppl. 5): S1-S59.
- Turillazzi E, Perilli G, Di Paolo M, Neri M, Riezzo I, Fineschi V. Side effects of AAS abuse: an overview. Mini Rev Med Chem 2011; 11: 374-389.
- Bhasin S, Woodhouse L, Casaburi T, et al. Testosterone dose-response relationships in healthy young men. Am J Physiol Endocrinol Metab 2001; 281: E1172-E1181.
- Van Zyl CG, Noakes TD, Lambert MI. Anabolic-androgenic steroid increases running endurance in rats. Med Sci Sports Exerc 1995; 27: 1385-1389.
- Georgieva KN, Boyadjiev NP. Effects of nandrolone decanoate on VO2max, running economy, and endurance in rats. Med Sci Sports Exerc 2004; 36: 1336-1341.
- Baume N, Schumacher YO, Sottas PE, et al. Effect of multiple oral doses of androgenic anabolic steroids on endurance performance and serum indices of physical stress in healthy male subjects. Eur J Appl Physiol 2006; 98: 329-340.
- Delgado J, Saborido A, Megias A. Prolonged treatment with the anabolic-androgenic steroid stanozolol increases antioxidant defences in rat skeletal muscle. J Physiol Biochem 2010; 66: 63-71.
- Buckley WE, Yesalis CE, Friedl KE, Anderson WA, Streit AL, Wright JE. Estimated prevalence of anabolic steroid use among male high school seniors. JAMA 1988; 260: 3441-3445.
- Bahrke MS, Yesalis CE, Kopstein AN, Stephens JA. Risk factors associated with anabolic-androgenic steroid use among adolescents. Sports Med 2000; 29: 397-405.
- Santos-Silva A, Rebelo MI, Castro EM, et al. Leukocyte activation, erythrocyte damage, lipid profile and oxidative stress imposed by high competition physical exercise in adolescents. Clin Chim Acta 2001; 306: 119-126.
- Aucouturier J, Baker JS, Duche P. Fat and carbohydrate metabolism during submaximal exercise in children. Sports Med 2008; 38: 213-238.
- Benitez-Sillero JD, Perez-Navero JL, Tasset I, Guillen-Del Castillo M, Gil-Campos M, Tunez I. Influence of intense exercise on saliva glutathione in prepubescent and pubescent boys. Eur J Appl Physiol 2009; 106: 181-186.
- Lambertucci RH, Levada-Pires AC, Rossoni LV, Curi R, Pithon-Curi TC. Effects of aerobic exercise training on antioxidant enzyme activities and mRNA levels in soleus muscle from young and aged rats. Mech Ageing Dev 2007; 128: 267-275.
- Sadowska-Krepa E, Klapcinska B, Jagsz S, et al. High-dose testosterone propionate treatment reverses the effects of endurance training on myocardial antioxidant defenses in adolescent male rats. Cardiovasc Toxicol 2011; 11: 118-127.
- Langfort J, Budohoski L, Newsholme EA. Effect of various types of acute exercise and exercise training on the insulin sensitivity of rat soleus muscle measured in vitro. Pflugers Arch 1988; 412: 101-105.
- Langfort J, Czarnowski D, Pilis W, Wojcik B, Gorski J. Effect of various types of exercise training on 5'-nucleotidase and adenosine deaminase activities in rat heart: influence of a single bout of endurance exercise. Biochem Mol Med 1996; 59: 28-32.
- Langfort J, Baranczuk E, Pawlak D, et al. The effect of endurance training on regional serotonin metabolism in the brain during early stage of detraining period in the female rat. Cell Mol Neurobiol 2006; 26: 1327-1342.
- Ohkawa H, Ohishi N, Yagi K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 1979; 95: 351-358.
- Finaud J, Lac G, Filaire E. Oxidative stress. Relationship with exercise and training. Sports Med 2006; 36:327-358.
- Bilski J, Mazur-Bialy AI, Wierdak M, Brzozowski T. The impact of physical activity and nutrition on inflammatory bowel disease: the potential role of cross talk between adipose tissue and skeletal muscle. J Physiol Pharmacol 2013; 64: 143-155.
- Neu CM, Rauch F, Rittweger J, Manz F, Schoenau E. Influence of puberty on muscle development at the forearm. Am J Physiol Endocrinol Metab 2002; 283: E103-E107.
- Gordon CS, Derino AS, Krause MP, et al. Impaired growth and force production in skeletal muscles of young partially pancreatomized rats: a model of adolescent type 1 diabetic myopathy? PLoS One 2010; 5: e14032.
- Locke M, Noble EG, Atkinson BG. Inducible isoform of HSP70 is constitutively expressed in a muscle fiber type specific pattern. Am J Physiol 1991; 261: C774-C779.
- Morton JP, Kayani AC, McArdle A, Drust B. The exercise-induced stress response of skeletal muscle, with specific emphasis on humans. Sports Med 2009; 39: 643-662.
- Skidmore R, Gutierrez JA, Guerriero V Jr, Kregel KC. HSP70 induction during exercise and heat stress in rats: role of internal temperature. Am J Physiol 1995; 268: R92-R97.
- Gonzalez B, Hernando R, Manso R. Stress proteins of 70 kDa in chronically exercised skeletal muscle. Pflugers Arch 2000; 440: 42-49.
- Milne KJ, Noble EG. Exercise-induced elevation of HSP70 is intensity dependent. J Appl Physiol 2002; 93: 561-568.
- Moran M, Delgado J, Gonzalez B, Mango R, Megias A. Responses of rat myocardial antioxidant defences and heat shock protein HSP72 induced by 12 and 24-week treadmill training. Acta Physiol Scand 2004; 180: 157-166.
- Feuerbacher I, Prinzinger R. The effects of the male sex-hormone testosterone on body temperature and energy metabolism in male Japanese quail (Coturnix coturnix japonica). Comp Biochem Physiol 1981; 70A: 247-250.
- Sanchez-Alavez M, Alboni S, Conti B. Sex- and age-specific differences in core body temperature of C57B1/6 mice. Age 2011; 33: 89-99.
- Kohno H, Takahashi N, Shinohara T, et al. Receptor-mediated suppression of cardiac heat-shock protein 72 expression by testosterone in male rat heart. Endocrinology 2007; 148: 3148-3155.
- Morimoto RI, Kroeger PE, Cotto JJ. The transcriptional regulation of heat shock genes: a plethora of heat shock factors and regulatory conditions. EXS 1996; 77: 139-163.
- Ahn SG, Thiele DJ. Redox regulation of mammalian heat shock factor 1 is essential for Hsp gene activation and protection from stress. Genes Dev 2003; 17: 516-528.
- Picard M, Hepple RT, Burelle Y. Mitochondrial functional specialization in glycolytic and oxidative muscle fibers: tailoring the organelle for optimal function. Am J Physiol Cell Physiol 2012; 302: C629-C641.
- Delchev S, Georgieva K, Terzieva D, Koeva Y, Atanassova P. Changes in androgen receptor expression in the myocardium of rats after submaximal training and nadrolone decanoate treatment. CR Acad Bulg Sci 2012; 65: 127-134.
- Deschenes MR, Maresh CM, Armstrong LE, Covault J, Kraemer WJ, Crivello JF. Endurance and resistance exercise induce muscle fiber type specific responses in androgen binding capacity. J Steroid Biochem Mol Biol 1994; 50: 175-179.
- Bricout VA, Germain PS, Serrurier BD, Guezennec CY. Changes in testosterone muscle receptors: effects of an androgen treatment on physically trained rats. Cell Mol Biol (Noisy-le-grand) 1994; 40: 291-294.
- Vicencio JM, Ibarra C, Estrada M, et al. Testosterone induces an intracellular calcium increase by a nongenomic mechanism in cultured rat cardiac myocytes. Endocrinology 2006; 147: 1386-1395.
- Foradori CD, Weiser MJ, Handa RJ. Non-genomic actions of androgens. Front Neuroendocrinol 2008; 29: 169-181.
- Chambon C, Duteil D, Vignaud A, et al. Myocytic androgen receptor controls the strength but not the mass of limb muscles. Proc Natl Acad Sci USA 2010; 107: 14327-14332.
- Janero DR. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indices of lipid peroxidation and peroxidative tissue injury. Free Radic Biol Med 1990: 9: 515-540.
- Storlien LH, Pan DA, Kriketos AD, et al. Skeletal muscle membrane lipids and insulin resistance. Lipids 2006; 31(Suppl.): S261-S265.
- Majerczak J, Rychlik B, Grzelak A, et al. Effect of 5-week moderate intensity endurance training on the oxidative stress, muscle specific uncoupling protein (UCP3) and superoxide dismutase (SOD2) contents in vastus lateralis of young, healthy men. J Physiol Pharmacol 2010; 61: 743-751.
- Chainy GB, Samantaray S, Samanta L. Testosterone-induced changes in testicular antioxidant system. Andrologia 1997; 29: 343-349.
- Pansarasa O, D'Antona G, Gualea MR, Marzani B, Pellegrino MA, Marzatico F. "Oxidative stress": effects of mild endurance training and testosterone treatment on rat gastrocnemius muscle. Eur J Appl Physiol 2002; 87: 550-555.
- Aydilek N, Aksakal M, Karakilcik AZ. Effects of testosterone and vitamin E on the antioxidant system in rabbit testis. Andrologia 2004; 36: 277-281.
- Liehr JG. Androgen-induced redox changes in prostate cancer cells: what are cause and effects? J Natl Cancer Inst 1997; 89: 3-4.
- Ripple MO, Henry WF, Rago RP, Wilding G. Prooxidant-antioxidant shift induced by androgen treatment of human prostate carcinoma cells. J Natl Cancer Inst 1997; 89: 40-48.
- Saartok T, Dahlberg E, Gustafsson JA. Relative binding affinity of anabolic-androgenic steroids: comparison of the binding to the androgen receptors in skeletal muscle and in prostate, as well as to sex hormone-binding globulin. Endocrinology 1984; 114: 2100-2106.
- Krieg M. Characterization of the androgen receptor in the skeletal muscle of the rat. Steroids 1976; 28: 261-274.
- Marsh JD, Lehmann MH, Ritchie RH, Gwathmey JK, Green GE, Schiebinger RJ. Androgen receptors mediate hypertrophy in cardiac myocytes. Circulation 1998; 98: 256-261.
- Langfort J, Jagsz S, Dobrzyn P, et al. Testosterone affects hormone-sensitive lipase (HSL) activity and lipid metabolism in the left ventricle. Biochem Biophys Res Commun 2010; 399: 670-676.
- Langfort J, Ploug T, Ihlemann J, Saldo M, Holm C, Galbo H. Expression of hormone-sensitive lipase and its regulation by adrenaline in skeletal muscle. Biochem J 1999; 340: 459-465.
- Ranatunga KW, Thomas PE. Correlation between shortening velocity, force-velocity relation and histochemical fibre-type composition in rat muscles. J Muscle Res Cell Motil 1990; 11: 240-250.
- Liu J, Yeo HC, Overvik-Douki E, et al. Chronically and acutely exercised rats: biomarkers of oxidative stress and endogenous antioxidants. J Appl Physiol 2000; 89: 21-28.
- Machida M, Takemasa T. Ibuprofen administration during endurance training cancels running-distance-dependent adaptations of skeletal muscle in mice. J Physiol Pharmacol 2010; 61: 559-563.
A c c e p t e d : October 8, 2013