BIOSYNTHESIS AND CATABOLISM OF MELATONIN
Melatonin is unique universal substance with the molecular structure unchanged throughout the animal and plant kingdom. It is produced in mammals, including human, mostly in the pineal gland, although several other organs (e.g., retina, extraorbital lacrimal gland, gastrointestinal tract, Harderian gland, bone marrow cells, blood platelets, and possibly other organs as well) may produce the hormone as well (3 - 5). Moreover, secretion of melatonin is not restricted to mammalian species but it is also produced in nonmammalian vertebrates, in some invertebrates, and in many plants (4, 6).
The synthesis of melatonin is presented in Fig. 1. The first step in melatonin formation is uptake of the amino acid L-tryptophan from the circulation into the gland. Within the pinealocyte tryptrophan-5-hydroxylase (L-tryptophan, tetrahydropteridine: oxygen oxidoreductase, EC 1.14.16.4) catalyzes L-tryptophan to 5-hydroxytryptophan which is then decarboxylated by L-aromatic amino acid decarboxylase (aromatic L-aminoacid carboxylase, EC 4.1.1.28) to serotonin. The next step, i.e., N-acetylation of serotonin to N-acetylserotonin is completed by arylalkulamine N-acetyltransferase (acetyl CoA:aryl-amine N-acetyltransferase, EC 2.3.1.5), the key enzyme in melatonin synthesis. The final step in the pathway is the O-methylation of N-acetylserotonin to melatonin by hydroxyindole-O-methyltransferase (S-adenosyl-L-methionine:N-acetyl-serotonin-O-methyltranferase, EC 2.1.1.4) (4, 7, 8). Once synthesized, melatonin is not stored in pineal cells but is quickly released into the bloodstream (9). Beside the blood melatonin is also present in other body fluids, including saliva, cerebrospinal fluid, bile, semen, amniotic fluid. Mean endogenous melatonin production rates have been calculated to be about 30 µg per day (10). The half-life of melatonin in serum has been calculated by various authors between less than 30 and 60 minutes (4, 6, 11).
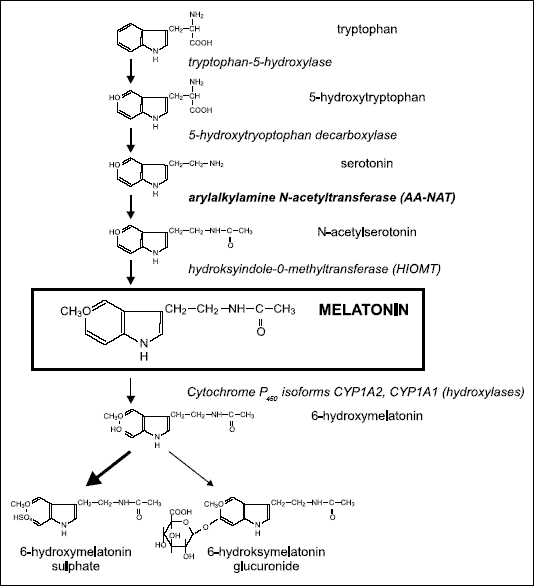 |
| Fig. 1. Synthetic pathway and metabolism of melatonin. |
Melatonin is metabolized primarily in the liver, and secondarily in the kidney. It undergoes 6-hydroxylation to 6-hydroxymelatonin, followed by sulfate or glucuronide conjugation to 6-hydroxymelatonin sulfate (90%) or 6-hydroxymelatonin glucuronide (10%) (Fig. 1). About 5% of serum melatonin content is excreted unmetabolized in urine. Melatonin forms also some minor metabolites, such as cyclic 2-hydroxymelatonin, N-gamma-acetyl-N-2-formyl-5-methoxykynurenamine and N-gamma-acetyl-5-methoxykynurenamine (4, 7).
MELATONIN CIRCADIAN RHYTHM AND ITS REGULATION
The synthesis of melatonin is strictly controlled by lighting conditions. Photosensory information arrives at the pineal via polyneuronal pathway that begins in the retina and involves retinohypothalamic tract, suprachiasmatic nuclei, paraventricular nuclei, medial forebrain bundle, reticular formation, intermediolateral cell column of the spinal cord, superior cervical ganglia, internal carotid nerve, and nervii conarii (7, 11). Postganglionic sympathetic nerve fibers that ends at the pineal gland releases noradrenalin, which plays crucial role in the control of melatonin synthesis. Noradrenalin binds to pinealocyte ß-adrenergic receptors (and partially alpha-adrenergic receptors), activating adenylate cyclase through GTP-binding protein in the cell membrane, and increase cAMP levels leading to stimulation of the activity of N-acetyltransferase, and subsequently to synthesis of melatonin. Stimulation of alpha-adrenergic receptors potentates the ß-stimulation, and in this mechanism participate calcium ions, phosphatidylinositol, diacylglicerol, and protein kinase C (12).
Melatonin has a well-defined circadian rhythm with peak in its production in the pineal gland occurring during the daily dark period (80% of melatonin is synthesized at night) (Fig. 2). Melatonin is present in all living organisms from plants, through animal kingdom to humans, and from unicellular algae to man shows this characteristic circadian rhythm.
 |
| Fig. 2. Circadian profiles of serum melatonin concentrations in humans; gray area = period of darkness. |
Rhythm of melatonin synthesis/secretion is generated by the circadian pacemaker (oscillator, biological clock) situated in the suprachiasmatic nucleus (SCN) of the hypothalamus, and synchronized to 24 hours primarily by the light-dark cycle acting via the SCN. During the day serum concentrations of the hormone are low (10-20 pg/ml), significantly increase at night (80-120 pg/ml) with peak between 24:00 and 03:00 h. The onset of secretion is usually around 21:00-22:00 h and the offset at 07:00-09:00 h. Very close relationship to melatonin rhythm shows its major urinary metabolite 6-sulfatoxymelatonin (7).
The rhythm in melatonin concentrations appears in humans soon after birth, in 6-8 week of life, and seems to be well established in 21-24 week of life (13). Amplitude of the nocturnal peak in melatonin secretion reaches the highest levels between 4th and 7th year of age. There is a drop in melatonin concentrations around maturation, values remain relatively stable until 35-40 years, and thereafter diminish gradually reaching around 70s levels similar to daytime concentrations (7, 11, 14). As a consequence, in advanced age many individuals do not exhibit a day-night differences in melatonin secretion (Fig. 3).
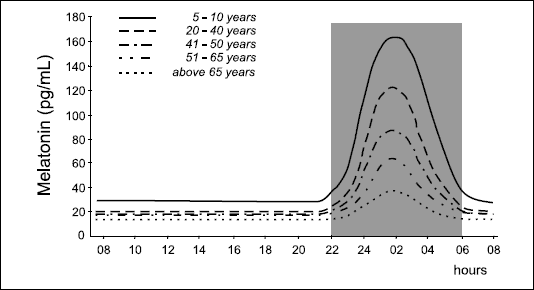 |
| Fig. 3. Circadian profiles of serum melatonin concentrations in humans at various age; gray area = period of darkness. |
Melatonin synthesis is rapidly suppressed in the dark phase by acute exposure to light of sufficient intensity, although there are substantial individual variations in human sensitivity to light among individuals that may be both genetically and environmentally determined (7).
The amplitude of nocturnal melatonin secretion is believed to be genetically determined and shows great differences among individuals (15). Thus, some individuals produce significantly less melatonin during lifetime than others. However, the circadian profile of melatonin has been found highly reproducible over a six-week period in the same subject (16).
MELATONIN RECEPTORS
Melatonin acts directly on target tissues through specific binding sites which are situated in the plasma membrane and nucleus of cells. According to the newest classification of nomenclature committee of IUPHAR, the best characterized and the most specific binding sites of melatonin are MT1 and MT2 membrane receptors belonging to the G-protein coupled receptor family (17). These receptors show similar high affinity to 2-(125I)-iodomelatonin radioligand but have different molecular structure and chromosomal gene localization. In humans MT1 receptor is mapped to chromosome 4q35.1 and consists of 350 amino acids (18). The gene for MT2 receptor is located into chromosome 11q21-22 and cDNA encodes a protein containing 363 amino acids (60% homology to MT1) (19). Melatonin binding with MT1 modulates intracellular signal via inhibiting adenylate cyclase and stimulating inositol phosphate (20). Activation of MT2 receptor inhibits formation of two second messengers cAMP and cGMP in cells (21). Third membrane receptor named MT3 is less known. Recently it has been shown that its structure is in 95% similar with human quinone reductase 2 and MT3 receptor participates in the regulation of intraocular pressure (22, 23). In mammals, the high-affinity melatonin receptors are found in the brain, mainly in hypothalamus and also in the pars tuberalis of hypophyseal. The pineal hormone acting through MT1 and MT2 receptors regulates the circadian rhythms and seasonal breeding of animals (17). Biological role of human membrane receptors have not been fully recognized. In human brain MT1 receptors are expressed in the suprachiasmatic nucleus, cerebellum, thalamus, hippocampus and cerebral cortex (24). MT2 mRNA has been presented in the human cerebellum and hippocampus (19, 25). Among other functions of melatonin, the neuroprotective action of hormone is postulated. It was shown that melatonin levels in Alzheimers disease (AD) patients are reduced and the in vitro study showed that melatonin prevents the human brain cells from amyloid ß-induced degeneration (26, 27). Neuroprotective effect of melatonin depends mainly on antioxidant activity. However, the receptor-mediated influence is possible, because MT1 and MT2 receptors were found in human hippocampus neurons. Moreover, it was observed that MT1 expression is higher and MT2 expression is lower in AD hippocampus (28, 29). MT1 gene expression and 2-(125I)-iodomelatonin binding were also found in following regions of human fetal brain: hypothalamus, thalamus, leptomeninges, cerebellum and brainstem (30). Melatonin of a pregnant woman easily crossing placenta can influence circadian rhythms of a fetus. Maternal melatonin and locally produced pineal hormone influences also via both membrane MT1 and MT2 receptors the function of the human placenta, and among others increases the hCG secretion from the trophoblast cells (31).
Melatonin receptors have been also discovered in several peripheral human tissues, including heart and arteries, kidney, liver, gallbladder, intestines, adipocytes granulosa cells of the ovarian follicle, uterus, breast cells, prostate and skin (32).
As a small lipophilic molecule, melatonin easily crosses cellular membranes and may also perform its biological function through cytoplasmatic and/or nuclear signaling. In 1994 the evidence of genomic action of melatonin via nuclear RZR/ROR receptors has been presented by Becker-Andre et al. (33). The RZR/ROR receptors belong to novel subclass of orphan nuclear receptors. They have been cloned simultaneously by two different groups and received the following names: retinoid Z receptor (RZR) and retinoid acid receptor-related orphan receptor (ROR) (34, 35). The RZR/ROR family consists of three subtypes: alpha, ß, and
The antitumor effect of melatonin is connected among others with antiproliferative and proapoptotic activities. The molecular mechanism of these actions still remains unclear, but several investigations have shown that oncostatic effects of melatonin may depend on membrane melatonin receptors and nuclear RZR/ROR receptors.
As a natural antioxidant, the pineal hormone should rather exhibit antiapoptotic properties. Indeed, several experiments involving mainly immune and neuronal cells have revealed the antiapoptotic action of melatonin (41 - 43). Recently, it has been found that melatonin may increase the apoptotis in tumor cells (44). The mechanism by which melatonin can induce apoptosis is unclear. The study conducted in our laboratory has shown that melatonin enhanced apoptosis in murine colonic cancer cells and nuclear RZR/ROR receptors agonist (CGP 52608) exerted a similar proapoptotic effects (45, 46). Moreover, we have found that thiazolidinedione CGP 55644 (an antagonist of nuclear RZR/RORalpha receptor) given together with melatonin diminishes its antiproliferative properties and blocks the proapoptotic effect of melatonin on colonic cancer cells (47) and completely blocks the inhibitory effects of melatonin on the growth of rat prolactin-secreting tumor (48).
In last years, the relationship between the estrogens and the antitumor action of melatonin has been the object of extensive investigations and the most of these studies have related to the breast cancer. It was shown that hormones growth-inhibitory effect reveals only in cancer cells having the estrogen receptors (49, 50). Melatonin interferes with estrogen receptor alpha (51). The antiestrogenic action of melatonin has been proposed to explain its oncostatic properties (52).
The investigations over the last years have shown that melatonin can modulate the immune system via both membrane and the nuclear receptors. The reduction of melatonin concentration in plasma causes a depression in humoral and cellular immune responses as well as inhibits the cytokines production (53). MT1 receptors and RZR/RORalpha receptors were identified in several human immunocompetent cells such as: monocytes, B lymphocytes, natural killer lymphocytes, T helper lymphocytes and cytotoxic T lymphocytes (54). In B lymphocytes melatonin binding to the RZR/RORalpha receptors down-regulates the expression of gene for 5-lipoxygenase, a important enzyme in allergic and inflammatory diseases like asthma and arthritis (55). The nuclear receptors involve also in cytokines secretion by human peripheral monocytes and cells of leukemia and lymphoma lines (56, 57).
Summing up, the results of many experimental studies strongly support the participation of MT1 and MT2 membrane receptors and nuclear RZR/ROR receptors in the action of melatonin. Moreover some evidence indicate that nuclear signaling plays an essential role in immunomodulatory and antitumor effects of pineal hormone.
MELATONIN IN HUMAN PHYSIOLOGY AND PATHOLOGY
Melatonin as an antioxidant
It has been discovered, recently, that melatonin is involved in antioxidative defense system of the organism, designed to protect molecules from damage by toxic oxygen radicals (58-60). Melatonin is a potent free radical scavenger and antioxidant that scavenges especially highly toxic hydroxyl radicals, and additionally stimulates a number of antioxidative enzymes. Because it is both lipophilic and hydrophilic, easily passes all morphophysiological barriers; enters all cells and may carry out its antioxidant function with equal efficiency in multiple cellular compartments, i.e. in the nucleus, cytosol and membranes (59). Moreover, it is the only antioxidant known to decrease substantially after middle age, and this decrease closely correlates with a decrease in total antioxidant capacity of human serum with age (61).
Question is still open, whether melatonin is efficient free radical scavenger also in physiological concentration or whether the observations made to date are of pharmacological importance only. However, it should be stressed that compared to two well-known scavengers, glutatione and mannitol, melatonin is 4x and 14x more effective, respectively (62). Free radical scavenging ability of melatonin has implications for variety of diseases, including age-associated neurodegenerative diseases and cancer initiation.
Melatonin and sleep and its disorders
There are many data (including those indicating the close relationship between the nocturnal increase of endogenous melatonin and the timing of sleep) suggesting involvement of melatonin in the physiological regulation of sleep (4, 63). Sleep promoting effects of melatonin have been well known since first experiments in early 70s, and is probably a consequence of increasing sleep propensity and of synchronizing effect on the circadian clock (64). The number of reports on melatonin concentrations in sleep disorders is surprisingly low considering its use in the therapy of insomnia. However, it has been demonstrated that the timing of the sleep gate was correlated with the onset of nocturnal melatonin secretion (65). Moreover, in fatal familiar insomnia (disease characterized by loss of sleep due to selective thalamic degeneration) serum melatonin concentrations gradually decrease as the disease progresses with complete rhythm obliteration in the most advanced stage (66).
Nocturnal melatonin concentrations were significantly lower in patients suffering from chronic primary insomnia (67, 68). In major sleep disorders such as narcolepsy, delayed sleep phase syndrome, and Klein Levine syndrome only a small delay in the melatonin rhythm was observed (7). Close association between the evening rise of melatonin levels and the evening increase in sleep propensity suggests a causal relationship (69, 70). Maximum melatonin secretion is also associated with nadir in alertness and performance as well as with maximum sleepiness/fatigue at night (69).
Lavie et al. (71) suggest that from the accumulated data it is evident that melatonin characteristics are not those of a typical hypnotic or sedative. Melatonin affects sleep in much more subtle way. The authors propose that the role of melatonin in the induction of sleep does not involve the active induction of sleep, but rather is mediated by an inhibition of a wakefulness-producing mechanism.
It has been demonstrated in several reports that administration of melatonin has beneficial effects in subjects (especially in advanced age) suffering from insomnia. In most recent reports melatonin was shown to significantly improve subjective and/or objective sleep parameters in some individuals. Its administration reduces sleep latency and/or increases sleep efficacy and total sleep time (64, 72, 73). Such effects are probably the consequence of increasing sleep propensity and of a synchronizing effect on the circadian clock (chronobiotic effect). However, we should keep in mind that melatonin is not a universally effective drug for treatment of insomnia, and it may not be helpful in all patients suffering from insomnia.
It should be noted, however, that although majority of data show that melatonin improve sleep parameters in elderly, in some studies sleep was unaffected by melatonin (see 64, 72 - 76). Moreover, two recent meta-analyses brought about different conclusions. Brzezinski et al. (77) concluded that melatonin is effective in increasing sleep efficiency and reducing sleep onset time whereas Buscemi et al. (78) failed to document clinically meaningful effects of egzogenous melatonin on sleep quality, efficiency or latency.
Melatonin and circadian rhytms and their disorders
Circadian rhythms play an important role in all living organisms. There are some indications of the relationship between melatonin and some body circadian rhythms. It is well known that in all mammalian species rhythmically produced melatonin (darkness hormone) functions as a photoperiodic signal and a circadian mediator, being one of critical components of internal biological clock(s) (79, 80). It is believed that melatonin could act as an endogenous synchronizer able to stabilize or to reinforce rhythms (81).
Wehr et al. (82) on the basis of the comparison between melatonin and other circadian rhythms proposed that temporal organization of the human circadian timing system exhibits distinct diurnal and nocturnal states with abrupt switch-like transitions between them. These states and transitions can be conceptualized as biological day and biological night and biological dawn and biological dusk. During biological day lack of melatonin secretion is accompanied by increasing core body temperature, decreasing sleepiness, decreasing wake EEG theta activity, decreasing REM sleep propensity, decreasing sleep propensity, and decreasing cortisol levels leading to wakefulness. On the contrary, during biological night melatonin secretion is accompanied by decreasing core body temperature, increasing sleepiness, increasing wake EEG theta activity, increasing REM sleep propensity, increasing sleep propensity, and increasing cortisol levels, leading to sleep.
There are many data suggesting a role of melatonin in circadian rhythm disorders. The jet-lag effect is perhaps the best clinical indication for melatonin use so far demonstrated (7, 64, 79). Air travelers well know that crossing several time zones during transcontinental flights causes many symptoms, including fatigue, sleepiness, irritability, apathy, digestive upsets, memory lapses, lack of concentration, impaired judgments and decision making, and headache (collectively known as jet-lag) causing distress to an increasing number of travelers. Majority of studies (both controlled and uncontrolled) indicate that melatonin administration is useful for ameliorating jet-lag symptoms (see 7, 64, 79). Moreover, the improvement is greater with the number of time zones, and in an eastwards direction compared to westwards (7).
In many blind people, especially in those with no conscious light perception and free running (non 24-h) rhythms, such circadian rhythm disorders as disrupted rhythms of sleep-wake cycle, core body temperature, cortisol, and melatonin are very common (83). Many blind subjects, have unusual melatonin or 6-sulfatoxymelatonin circadian profiles with the periodicity of the endogenous rhythm varying from 23h50min to 25h00min (84, 85). Melatonin has proven efficacy in phase-shifting of the circadian clock for phase resetting in blind people. It may stabilize sleep onset and sometimes improve quality and duration of sleep (7, 83, 86).
Circadian rhythms are also disturbed in shift workers (especially permanent night shift workers) who often complain of fatigue, sleep disturbances, and gastrointestinal problems (79). Great variability in circadian melatonin profiles, with the onset of the melatonin secretion varying between 21:45 h and 05:05 h, has been demonstrated in night workers (87). Melatonin, when administered at the desired bedtime during a night shift, may improve sleep and increase daytime alertness in shift workers, (7, 79), and therefore, may prove to be a useful strategy for helping real night workers adapt to working night shifts (88).
It seems that melatonin is the effective chronobiotic, i.e. a chemical substance capable of therapeutically re-entraining short-term dissociated or long-term desynchronized circadian rhythms, or prophylactically preventing disruption following environmental insult (89).
Melatonin and immune system
Many data, both from animal and human studies, point to immunomodulatory potential of melatonin (90-93). It has been demonstrated that such parameters of immune reactivity as number of immune cells and their subpopulations, lymphocyte proliferation, blood level of different cytokines, phagocytic index, etc., exhibit well pronounced circadian rhythmicity (94), and these diurnal changes in the immune system function seem to be controlled by or correlated with the pineal melatonin synthesis and secretion (95).
It seems that melatonin may exert a direct influence on the immune system because melatonin receptors (both membrane and putative nuclear) have been discovered in immune organs and cells of humans and various mammalian species (91). Moreover, it was recently reported that cultured human lymphocytes synthesize and release large amount of melatonin which could act, in addition to its endocrine effect, as an intracrine, autocrine, and/or paracrine substance for the local coordination of the immune response (96). Our recent data suggest that endogenous melatonin is an essential part for an accurate response of human lymphocytes through the modulation on interleukin-2/interleukin-2 receptor system (97).
However, the relationship between melatonin and immune system seems to be complex and needs further elucidation.
Melatonin and pituitary hormones
The data on the relationship between melatonin and pituitary hormones are inconsistent. There are some data suggesting the relationship between melatonin and prolactin. The diurnal concentrations of melatonin positively correlate with those of prolactin (98, 99), nocturnal increase, and morning decrease in prolactin levels are proceeded by similar changes in melatonin levels (100), and melatonin administration stimulates prolactin secretion (99, 101). Increased nocturnal serum melatonin concentrations or urinary 6-sulfatoxymelatonin excretion were found in majority of studies in hyperprolactinemic patients compared to their age-matched healthy individuals (102-104). Moreover, administration of 5 mg of melatonin in healthy women resulted in a rapid and prominent prolactin release, similar to that observed at nighttime in patients with hyperprolactinemia (102). However, it does not seem probable that melatonin plays important role in the control of prolactin secretion.
Relationship between melatonin and growth hormone (GH) is poorly understood. Decrease in melatonin concentrations has been observed following stimulation of GH (due to insulin-induced hypoglycemia, arginine infusion, clonidine administration, or growth hormone releasing hormone stimulation) in children (105). Moreover, our recent results showed that melatonin levels were significantly higher in children with GH deficiency in comparison with children with idiopathic short stature, and there was negative correlation between GH peak after stimulation test and nocturnal melatonin concentrations (106). Administration of melatonin caused either enhancement of spontaneous and exercise-induced GH secretion (107, 108), or did not exert any effect (101). However, role of melatonin in mechanisms of regulation of growth hormone secretion seems to be secondary and not important.
There are experimental data suggesting relationship between the pineal gland and hypothalamo-hypophysial-thyroid axis in animals, however, no sufficient data are available on the existence of such relationship in humans (109). Also, no relationship between melatonin and hypothalamic-pituitary-adrenal axis seems to exist (5, 8).
Melatonin and reproductive system
The relationship between the pineal and reproductive system is well established in animals indicating that melatonin regulates the reproduction in sesonally breading animals by its action at various levels of the hypothalamic-pituitary-gonadal axis (110). However, in humans it is more difficult to demonstrate, despite the fact that first association between pineal gland and reproductive system has been suggested in humans already in 19th century. Some studies suggest that melatonin may play a role in physiological development of normal puberty (105, 111). Precocious puberty or delayed puberty is often associated with abnormal melatonin levels (112, 113). Although there are no sufficient data indicating significant role of melatonin in puberty, it seems probable that differences in melatonin concentrations may be responsible for some subtle changes in secretion of gonadotropins or influence the mechanism of pulsatile GnRH secretion, and therefore affects sexual maturation.
Moreover, melatonin may mediate the moderate seasonal fluctuations observed in human reproductive function (4). Elevated concentrations of melatonin were reported in male infertility (114), and in men with hypogonadotropic hypogonadism (115, 116). On the contrary, in men with hypergonadotropic hypogonadism melatonin secretion is decreased, and is normalized following testosterone treatment (117). High nocturnal melatonin concentration was demonstrated also in women with hypothalamic amenorrhea (118, 119). Increase in urinary 6-sulfatoxymelatonin excretion was found also in hyperandrogenic women with polycystic ovary syndrome (120).
Melatonin in various pathologies
Alterations in melatonin concentrations and/or its circadian rhythm were found in various psychiatric disorders, such as major depression, bipolar affective disorder, panic disorder, obsessive compulsive disorder, schizophrenia, eating disorders, cluster headache, most conspicuously in the cluster period diabetic autonomic neuropathy, and in Smith-Magenis syndrome (see 5, 121-123). Lower nocturnal melatonin levels were observed in alcoholic patients as compared with control individuals. Moreover, depressed melatonin concentrations were observed even after long abstinence, suggesting that chronic use of alcohol might permanently alter the pineal ability to produce melatonin (124).
Altered circadian melatonin rhythm was also observed in several other pathologies like: liver cirrhosis, chronic renal failure both with compensated disease and in end-stage renal disease, psoriasis, duodenal ulcer, night-eating syndrome, cardiovascular diseases, and others (see 5).
Melatonin and neurodegenerative diseases
A role for melatonin in neurodegenerative diseases (such as Alzheimers and Parkinsons diseases) has been recently suggested.
The experimental findings indicate that melatonin may act in a variety of ways to reduce neuronal loss in Alzheimers disease by altering the process of generation and action of amyloid-ß leading to increased cellular survival. Melatonin concentrations decrease in some, but not all, patients suffering from Alzheimers disease. Decreased nocturnal melatonin levels were found both in preclinical and definite Alzheimers patients. Many reports demonstrated that melatonin treatment seems to constitute a selection therapy to improve sleep, to ameliorate sundowning, and to slow evolution of cognitive impairment in Alzheimers patients (see 125, 126).
There are also experimental data that suggest a role of melatonin in another neurodegenerative disorder, Parkinsons disease which is characterized by the progressive deterioration of dopamine-containing neurons in the pars compacta of the substantia nigra in the brain stem due to the oxidation of dopamine (127). There is evidence that melatonin may reduce dopamine auto-oxidation under experimental conditions (128) although its administration did not slow progression of the Parkinson disease (see 125).
Melatonin and neoplastic disease
Although the relationship between the pineal gland, melatonin, and neoplastic disease has been demonstrated in various experimentally-induced animal tumors, and in the majority of studies melatonin has been shown to inhibit development and/or growth of various experimental animal tumors and some human cell lines in vitro its role in human malignancy is not clear (reviewed in 129, 130). Hovewer, depressed nocturnal melatonin concentrations or nocturnal excretion of the main melatonin metabolite 6-sulfatoxymelatonin were found in various tumor types (breast cancer, prostate cancer, colorectal cancer, endometrial cancer, cervical cancer, lung cancer, and stomach cancer), whereas in other tumor types (Hodgkins sarcoma, osteosarcoma, ovarian cancer, laryngeal cancer, and urinary bladder cancer) melatonin levels were not changed or showed great variations among individuals (5, 131).
Moreover, some clinical studies performed mainly by Lissonis group suggest that administration of melatonin (in relatively high doses either alone or in combination with IL-2) is able to favorably influence the course of advanced malignant disease in humans and lead to an improvement in their quality of life (reviewed in 131-133). However, these observations require to be verified by independent and controlled studies.
Melatonin and aging
Rapid increase of the size of the elderly population (over the age 65), both in numbers and as a proportion of the whole raises many social and economic problems because these beneficiaries of health and pension funds are supported by a relatively smaller number of potential contributors in the economically active age, and results also increase in number of people suffering from age-related diseases (such as atherosclerosis, neoplastic disease, neurodegenerative diseases). Therefore, there is a search for any therapeutic agent improving quality of life of elderly. A role for melatonin as such a compound was recently suggested.
Although many theories relating melatonin to aging have been put forward, the role of this compound in the aging process is not clear. Aging is beyond a doubt mulfifactorial process, and no single element seems to be of basic importance. Although there is not clear evidence indicating that melatonin may delay aging there are some reasons to postulate a role for this compound in the aging process: (i) melatonin participates in many vital life processes, and its secretion falls gradually over the life-span; (ii) diminished melatonin secretion in advanced age may be related to deterioration of many circadian rhythms, as a consequence of a reduced function of suprachiasmatic nucleus; (iii) Melatonin acts as endogenous sleep-inducing agent, and its reduced concentrations may result in lowered sleep efficacy very often associated with advancing age; (iv) melatonin exhibits immunoenhancing properties, and suppressed immunocompetence has been implicated in the acceleration of aging processes; (v) melatonin is a potent free radical scavenger, and free radicals cause damage to vital cellular constituents, accumulating with age which has significance not only for aging per se but also for many age-related diseases (4, 14, 134).
Aging is beyond a doubt mulfifactorial process, and no single element seems to be of basic importance. However, the age-related decline in melatonin secretion may have various consequences including sleep inefficiency, circadian rhythm dysregulation, depressed immune function, reduced antioxidant protection, and possibly others (14). Recent findings of Kunz et al. (135) show that exogenous melatonin, when administered at the appropriate time, seems to normalize circadian variation in human physiology, and therefore, melatonin may have impact on general health, especially in the elderly.
Possible therapeutic significance of melatonin
It has been proposed that melatonin may be of some therapeutic significance. Moreover, in some countries (e.g. Argentina, China, Poland, USA) melatonin has become recently available as either an OTC drug or food supplement. There are some widely accepted indications for therapeutic use of melatonin but also perspectives for its broader use (136).
Generally, melatonin has been proven to be useful in circadian rhythm disorders, such as sleep disturbances, jet lag, sleep-wake cycle disturbances in blind people, and shift work. Other possibilities for therapeutic usefulness of melatonin are not definitively proved.
It should be stressed that toxicity of melatonin is remarkably low, and no serious negative side effects of melatonin have been reported, so far (136).
- Lerner AB, Case JD, Takahashi Y, Lee TH, Mori N. Isolation of melatonin, pineal factor that lightens melanocytes. J Am Chem Soc 1958; 80: 2587.
- Lerner AB, Case JD, Heinzelman RU. Structure of melatonin. J Am Chem Soc 1959; 81: 6084-6085.
- Vijayalaxmi, Thomas RC, Reiter RJ, Herman TS. Melatonin: from basic research to cancer treatment clinics. J Clin Oncol 2002; 20: 2575-2601.
- Pandi-Perumal SR, Srinivassan V, Maestroni GJM, Cardinali DP, Poeggeler B, Harderland R. Melatonin. Natures most versatile biological signal? FEBS J 2006; 273: 2813-2836.
- Karasek M. Melatonin in human physiology and pathology. In Frontiers in Chronobiology Research, F Columbus (ed). Hauppage, NY, Nova Science, 2006, pp. 1-43.
- Harderland R, Poeggeler B. Non-vertebrate melatonin. J Pineal Res 2003; 34: 233-241.
- Arendt J. Melatonin and the mammalian pineal gland. London, Chapman & Hall, 1995.
- Karasek M. Melatonin in humans where we are 40 years after its discovery. Neuroendocrinol Lett 1999; 20: 179-188.
- Reiter RJ. Melatonin: clinical relevance. Best Pract Res Clin Endocrinol Metab 2003; 17: 273-285.
- Lane EA, Moss HB. Pharmacokinetics of melatonin in man: first pass hepatic metabolism. J Clin Endocrinol Metab 1985; 61: 1214-1216.
- Brown EN, Choe Y, Shanahan TL, Czeisler CA. A mathematical model of diurnal variations in human plasma melatonin levels. Am J Physiol 1997; 272: E506-516.
- Sugden D. Melatonin biosynthesis in the mammalian pineal gland. Experientia 1989; 45: 922-931.
- Kennaway DJ, Stamp GE, Goble FC. Development of melatonin production in infants and the impact of prematurity. J Clin Endocrinol Metab 1992; 75: 367-369.
- Karasek M. Melatonin, human aging, and age-related diseases. Exp Gerontol 2004; 39: 1723-1729.
- Bergiannaki JD, Soldatos CR, Paparrigopoulos TJ, Syrengelas M, Stefanis CN. Low and high melatonin excretors among healthy individuals. J Pineal Res 1995; 18: 159-164.
- Selmaoui B, Touitou Y. Reproducibility of the circadian rhythm of serum cortisol and melatonin in healthy subjects: a study of three different cycles over six weeks. Life Sci 2003; 73: 3339-3349.
- Dubocovich ML, Cardinali DP, Delagrange P, et al. Melatonin receptors. In The IUPHAR Compendium of Receptor Characterization and Classification, 2nd edition, IUPHAR Media, London, UK, 2000, pp. 270-277.
- Slaugenhaupt SA, Roca AL, Liebert CB, Altherr MR, Gusella JF, Reppert SM. Mapping of the gene for the Mel1a melatonin receptor to human chromosome 4 (MTNR1A) and mouse chromosome 8 (Mtnr1a). Genomics 1995; 27: 355-357.
- Reppert SM, Godson C, Mahle CD, Weaver DR, Slaugenhaupt SA, Gusella JF. Molecular characterization of second melatonin receptor expressed in human retina and brain: the Mel1b melatonin receptor. Proc Natl Acad Sci USA 1995; 92: 8734-8738.
- Godson C, Reppert SM. The Mel1a melatonin receptor is coupled to parallel signal transduction pathways. Endocrinol 1997; 138: 397-404.
- Petit L, Lacroix I, Coppet P, Strosberg AD, Jockers R. Differential signaling of human Mel1a and Mel1b melatonin receptors trough the cyclic guanosine 3-5-momphosphate pathway. Biochem Pharmacol 1999; 58: 633-639.
- Nosjean O, Ferro M, Coge F, et al. Identification of the melatonin-binding site MT3 as the quinone reductase 2. J Biol Chem 2000; 275: 31311-31317.
- Pintor J, Martin L, Pelaez T, Hoyle CH, Peral A. Involvement of melatonin MT(3) receptors in the regulation of intraocular pressure in rabbits. Eur J Pharmacol 2001; 416: 251-254.
- Mazzuchelli C, Pannacci M, Nonno R, Lucini V, Fraschini F, Stankow BM. The melatonin receptor in the human brain: cloning experiments and distribution studies. Mol Brain Res 1996; 39: 117-126.
- Al-Ghoul WM, Herman MD, Dubocovich ML. Melatonin receptor subtype expression in human cerebellum. Neuroreport 1998; 9: 4063-4068.
- Ferrari E, Arcaini A, Gornati R, et al. Pineal and pituitary-adrenocortical function in physiological aging and in senile dementia. Exp Gerontol 2000; 35: 1239-1250.
- Pappolla MA, Chyan YJ, Poeggeler B, et al. An assessment of the antioxidant and the antiamyloidogenic properties of melatonin: implications for Alzheimers amyloid disease. J Neural Transm 2000; 107: 203-231.
- Savaskan E, Olivieri G, Meier F, et al. Increased melatonin 1a-receptor immunoreactivity in the hippocampus of Alzheimers disease patients. J Pineal Res 2002; 31: 59-62.
- Savaskan E, Ayoub MA, Ravid R, et al. Reduced hippocampal MT2 melatonin receptor expression in Alzheimers disease. J Pineal Res 2005; 38: 10-16.
- Thomas L, Purvis CC, Drew JE, Abramovich DR, Williams LM. Melatonin receptors in human fetal brain: 2-[125I]iodomelatonin binding and MT1 gene expression. J Pineal Res 2002; 33: 218-224.
- Iwasaki S, Nakazawa K, Sakai J, et al. Melatonin as a local regulator of human placental function. J Pineal Res 2005; 39: 261-265.
- Ekmekcioglu C. Melatonin receptors in humans: biological role and clinical relevance. Biomed Pharmacother 2006; 60: 97-108.
- Becker-Andre M, Wiesenberg I, Schaeren-Wiemers N, et al. Pineal gland hormone melatonin binds and activates an orphan of the nuclear receptor superfamily. J Biol Chem 1994; 269: 28531-28534.
- Becker-Andre M, Andre E, De Lamarter JF. Identification of nuclear receptor mRNAs by RT-PCR amplification of conserved zinc-finger motif sequences. Bioch Biophys Res Commun 1993; 194: 1371-1379.
- Giguere V, Tini M, Flock G, Ong E, Evans RM, Otulakowski G. Isoform-specific amino-terminal domains dictate DNA-binding properties of RORa, a novel family of orphan hormone nuclear receptors. Gen Dev 1994; 8: 538-553.
- Giguere V. Orphan nuclear receptors: from gene to function. Endocrine Rev 1999; 20: 689-725.
- Karasek M; Carrillo-Vico A; Guerrero JM, Winczyk K, Pawlikowski M. Expression of melatonin MT1 and MT2 receptors, and RORalpha1 receptor in transplantable murine Colon 38 cancer. Neuroendocrinol Lett 2002; 23(suppl 1): 55-60.
- Moretti RM, Montagnani Marelli M, Motta M, Limonta P. Oncistatic activity of a thiazolidinedione derivative on human androgen-dependent prostate cancer cell. Int J Cancer 2001; 92: 733-737.
- Moretti RM, Montagnanii Marelli M, Motta M, et al. Activation of the orphan nuclear receptor RORalpha induces growth arrest in androgen-independent DU 145 prostate cancer cells. Prostate 2001; 46: 327-335.
- Dai J, Ram PT, Yaun L, Spriggss LL, Hill SM. Transcriptional repression of RORa, activity in human breast cancer cells by melatonin. Mol Cell Endocrinol 2001; 176: 111-120.
- Provinciali M, Di Stefano G, Bulina D, Tibaldi A, Fabris N. Effect of melatonin and pineal grafting on thymocytes apoptosis in aging mice. Mech Ageing Dev 1996; 90: 1-19.
- Cagnoli CM, Atabay C, Kharfamova E, Manew H. Melatonin protects neurons from singlet oxygen-induced apoptosis. J Pineal Res 1995; 18: 222-226.
- Deigner HP, Haberkorn U, Kinscherf R. Apoptosis modulators in the therapy of neurodegenerative diseases. Exp Opin Investig Drugs 2000; 9: 747-764.
- Sainz RM, Mayo JC, Rodriguez, Tan DX, Lopez-Burillo S, Reiter RJ. Melatonin and cell death: differential actions on apoptosis in normal and cancer cells. Cell Mol Life Sci 2003; 60: 1407-1426.
- Melen-Mucha G, Winczyk K, Pawlikowski M. Somatostatin analogue octreotide and melatonin inhibit bromodeoxyuridine incorporation into cell nuclei and enhance apoptosis in the transplantable muurine Colon 38 cancer. Anticancer Res 1998; 18: 3615-3620.
- Winczyk K, Pawlikowski M, Karasek M. Melatonin and RZR/ROR receptor ligand CGP 52 608 induce apoptosis in the murine colonic cancer. J Pineal Res 2001; 31: 179-182.
- Winczyk K, Pawlikowski M, Guerrero JM, Karasek M. Possible involvement of the nuclear RZR/ROR-alpha receptor in the antitumor action of melatonin on murine Colon 38 cancer. Tumor Biol 2002; 23: 298-302.
- Karasek M, Gruszka A, Lawnicka H, Kunert-Radek J, Pawlikowski M. Melatonin inhibits growth of diethylstilbestrol-induced prolactin-secreting pituitary tumor in vitro: possible involvement of nuclear RZR/ROR receptors. J Pineal Res 2003; 34: 1-3.
- Ram PT, Dai J, Yuan L, et al. Involvement of the MT1 melatonin receptor in human breast cancer. Cancer Lett 2002; 179: 141-150.
- Kanishi Y, Kobayashi Y, Noda S, Ishizuka B, Saito K. Differential growth inhibitory effect of melatonin on two endometrial cancer cell lines. J Pineal Res 2000; 28: 227- 233.
- Dai J, Inscho EW, Yuan L, Hill SM. Modulation of intracellular calcium and calmodulin by melatonin in MCF-7 human breast cancer cells. J Pineal Res 2002; 32: 112-119.
- Sánchez-Barcelo EJ, Cos S, Mediavilla D, Martinez-Campa G, Alonso-González C. Melatonin-estrogen interactions in breast cancer. J Pineal Res 2005; 38: 217-222.
- Carrillo-Vico A, Guerrero JM, Lardone PJ, Reiter RJ. A review of the multiple actions of melatonin on the immune system. Endocrine 2005; 27: 189-200.
- Pozo D, Garcia-Maurino S, Guerrero JM, Calvo JR. mRNA expression of nuclear RZR/ROR alpha, membrane receptor MT, and hydroxyindole-O-methyltransferase in different populations of human immune cells. J Pineal Res 2004; 37: 48-54.
- Steinhilber D, Brungs M, Werz O et al. The nuclear receptor for melatonin represses 5-lipoxygenase gene expression in human B lymphocytes. J Biol Chem 1995; 270: 7040-7073.
- Garcia-Maurino S, Gonzalez-Haba MG, Calvo JR, Goberna R, Guerrero JM. Involvement of nuclear binding sites for melatonin in the regulation of IL-2 and IL-6 production by human blood mononuclear cells. J Neuroimmunol 1998; 92: 76-84.
- Garcia-Maurino S, Pozo S, Calvo JR, Guerrero JM. Correlation between nuclear melatonin receptor expression and enhanced cytokine production in human lymphocytic and monocytic cell lines. J Pineal Res 2000; 29: 129-137.
- Reiter RJ. Oxygen radical detoxification processes during aging: The functional importance of melatonin. Aging Clin Exp Res 1995; 7: 430-351.
- Reiter RJ. Oxidative damage in the central nervous system: protection by melatonin. Progr Neurobiol 1998, 56, 359-384.
- Reiter, RJ, Chen, L-D, Poeggeler, BH, Barlow-Walden, L, Sewerynek, E, Melchiori, D. Antioxidant actions of melatonin and related compounds. In Handbook of Antioxidants, E Cadenas, L Packer (eds). New York, Marcel Dekker, 1996b, pp. 513-541.
- Benot S, Goberna R, Reiter RJ, Garcia-Maurino E, Osuna C, Guerrero, JM. Physiological levels of melatonin contribute to the antioxidant capacity of human serum. J Pineal Res 1999; 27: 59-64.
- Tan DX, Chen LD, Poeggeler B, Manchester LC, Reiter RJ. Melatonin: A potent, endogenous hydroxyradical scavenger. Endocrine J. 1993; 1: 57:60.
- Zhdanova IV. Melatonin as a hypnotic: pro. Sleep Med Rev 2005; 9: 51-65.
- Cardinali DP, Brusco LI, Perez Lloret S, Furio AM. Melatonin in sleep disorders and jet-lag. Neuroendocrinol Lett 2002; 23 (suppl 1): 9-13.
- Tzischinsky O, Shlitner A, Lavie P. The association between the nocturnal sleep gate and nocturnal onset of urinary 6-sulphatoxymelatonin. J Biol Rhythms 1993; 8: 199-209.
- Portaluppi F, Cortelli P, Avoni P et al. Progressive disruption of the circadian rhythm of melatonin in fatal familiar insomnia. J Clin Endocrinol Metab 1994; 78: 1075-1078.
- Hajak G, Rodenbeck A, Staedt J, Bandelow B, Huether G, Rüther E. Nocturnal plasma melatonin levels in patients suffering from chronic primary insomnia. J Pineal Res 1995; 19: 116-122.
- Riemann D, Klein T, Rodenbeck A, et al. Nocturnal cortisol and melatonin secretion in primary insomnia. Psychiatry Res 2002; 113: 17-27.
- Arendt J. Importance and relevance of melatonin to human biological rhythms. J Neuroendocrinol 2003; 15: 427-431.
- Lavie, P. Melatonin: role in gating nocturnal rise in sleep propensity. J Biol Rhythms 1997; 12: 657-665.
- Lavie P, Haimov I, Shochat T. Melatonin shutting off the wakefulness system. Front Horm Res 1997; 23: 149-160.
- Zisapel N. The use of melatonin for the treatment of insomnia. Biol Signals Recept 1999; 9: 84-89.
- Monti JM, Cardinali DP. A critical assessment of the melatonin effect on sleep in humans. Biol Signals Recept 2000; 9: 328-339.
- Skene D, Lockley SW, Arendt J. Use of melatonin in the treatment of phase shift and sleep disorders. Adv Exp Med Biol 1999; 467: 79-84.
- Pandi-Perumal SR, Seils LK, Kayumov L, Ralph MR, Lowe A, Moller H, Swaab DF. Senescence, sleep, and circadian rhythms. Aging Res Rev 2002; 1: 559-604.
- Baskett JJ, Broad JB, Wood PC, et al. Does melatonin improve sleep in older people? A randomized crossover trial. Age Ageing 2003; 32: 164-170.
- Buscemi N, Vansermeer B, Hooton N, et al. Efficacy and safety of endopgenous melatonin for secondary dleep disorders and sleep disorders accompanying sleep restriction: meta-analysis. BMJ 2006; 332: 385-393.
- Brzezinsky A, Vangel MG, Wurtman RJ, et al. Effects of endogenous melatonin on sleep: a meta-analysis. Sleep Med Rev 2005; 9: 41-50.
- Arendt J, Deacon S. Treatment of circadian rhythm disorders melatonin. Chronobiol Int 1997; 14: 185-204.
- Pevet P. Melatonin and biological rhythms. Biol Signals Recept 2000; 9: 203-212.
- Armstrong SM. Melatonin: the internal zeitgeber of mammals? Pineal Res Rev 1989; 7: 157-202.
- Wehr TA, Aeschbach D, Duncan WC Jr. Evidence for a biological dawn and dusk in the human circadian timing system. J Physiol 2001; 535: 937-851.
- Skene DJ, Lockley SW, Arendt J. Melatonin in circadian sleep disorders in the blind. Biol Signals Recept 1999; 8: 90-95.
- Lewy AJ, Newsome DA. Different types of melatonin circadian secretory rhythms in some blind people. J Clin Endocrinol Metab 1983; 56: 1103-1107.
- Lockley SW, Skene DJ, Arendt J Tabandeh H, Bird AC, Defrence R. Relationship between melatonin rhythms and visual loss in the blind. J Clin Endocrinol Metab 1997; 82: 3763-3770.
- Lockley SW, Skene DJ, James K, Thapan K, Wright J, Arendt, J. Melatonin administration can entrain the free-running circadian system in blind subjects. J Endocrinol 2000; 164: R1-6.
- Wiebel L, Spiegel K, Gronfier C, Follenius M, Brandenberger G. Twenty-four-hour melatonin and core body temperature rhythms: their adaptation in night workers. Am J Physiol 1997; 272: R948-954.
- Sharkey KM, Eastman CI. Melatonin phase shifts human circadian rhythms in a placebo-controlled simulated night-work study. Am J Physiol Regul Integr Comp Physiol 2002; 282: R454-463.
- Dawson D, Armstrong SM. Chronobiotics drugs taht shift rhythms. Pharmacol Ther 1996; 69: 15-36.
- Maestroni GJM. The immunotherapeutic potential of melatonin. Exp Opin Invest Drugs 2001; 10: 467-476.
- Guerrero JM, Reiter RJ. Melatonin-immune system relationships. Curr Top Med Chem 2002, 2, 167-179.
- Carrillo-Vico A, Guerrero JM, Lardone PJ, Reiter RJ. A review of the multiple actions of melatonin on the immune system. Endocrine 2005; 27: 189-200.
- Pandi-Perumal SR, Esquifino AI, Cardinali DP, Miller SC, Maestroni GJM. The role of melatonin in immunoenhamcement: potential application in cancer. Int J Exp Pathol 2006; 87: 81-87.
- Petrovsky N, Harrison LC. Diurnal rhythmicity of human cytokine production. J Immunol 1997; 158: 5163-5168.
- Nelson RJ, Drazen DL. Melatonin mediates seasonal changes in immune function. Ann N Y Acad Sci 2000; 917: 404-415.
- Carrillo-Vico A, Calvo JR, Abreu P, et al. Evidence of melatonin synthesis by human lymphocytes and its physiological significance: possible role as intracrine, autocrine, and/or paracrine substance. FASEB J 2004; 18: 537-539.
- Carrillo-Vico A, Lardone PJ, Fernandez-Santos JM, et al. Human lymphocyte-synthesized melatonin is involved in the regulation of the IL-2/IL-2 receptor system. J Clin Endocrinol Metab 2005, 90, 992-1000.
- Rao ML, Mager T. Influence of the pineal gland on pituitary function in humans. Psychoneuroendocrinology 1987; 12: 141-147.
- Webley GE, Böhle A, Leidenberger F. Positive relationship between nocturnal concentrations of melatonin and prolactin, and a stimulation of prolactin after melatonin administration in young men. J Pineal Res 1988; 5: 19-23.
- Webley GE, Lenton EA. The temporal relationship between melatonin and prolactin in women. Fertil Steril 1987; 48: 218-222.
- Waldahauser F, Lieberman HR, Lynch HJ, et al. A pharmacological dose of melatonin increases PRL levels in males without altering those of GH, LH, FSH, TSH, testosterone or cortisol. Neuroendocrinology 1987; 46: 125-130.
- Okatani Y, Okada M, Sagara Y. Amplification of nocturnal melatonin secretion in women with nocturnal hyperprolactinemia. Asia Oceania J Obstet Gynecol 1992; 18: 289-297.
- Blaicher W, Imhof MH, Gruber DM, Schneeberger C, Sator MO, Huber JC. Endocrinological disorders. Focusing on melatonins interactions. Gynecol Obstet Invest 1999; 48: 179-182.
- Karasek M, Stawerska R, Hilczer M, Zylinska K, Lewinski A. Melatonin circadian rhythm in women with idiopathic hyperprolactinemia. Neuroendocrinol Lett 2004, 25, 411-414.
- Gupta D, Attanasio A. Pathophysiology of pineal function in health and disease in children. Pineal Res Rev 1988; 6: 261-300.
- Karasek M, Stawerska R, Smyczynska J, Lewinski A. Increased melatonin concentrations in children with growth hormone deficiency. J Pineal Res, in press.
- Valcavi R, Dieguez C, Azzarito C, et al. Effect of oral administration of melatonin on GH responses to GRF 1-44 in normal subjects. Clin Endocrinol 1987; 26: 453-458.
- Meeking DR, Wallace JD, Cuneo RC, Forsling M, Russel-Jones DL. Exercise-induced GH secretion is enhanced by the oral ingestion of melatonin in healthy adult male subjects. Eur J Endocrinol 1999; 141: 22-26.
- Lewinski A, Karbownik M. Melatonin and the thyroid gland. Neuroendocrinol Lett 2002; 23(suppl 1): 73-78.
- Reiter RJ. Mechanisms of control of reproductive physiology by the pineal gland and its hormones. Adv Pineal Res 1987; 2: 109-125.
- Lang U. Melatonin and puberty. Pineal Res Rev 1986; 4: 199-243.
- Gupta, D. Human development and the pineal gland. In The Pineal Gland During Development: From Fetus To Adult, D Gupta, RJ Reiter (eds). London, Croom Helm, 1986, pp. 117-133.
- Waldhauser F, Boepple PA, Cowley WF Jr. Low nocturnal serum melatonin levels in precocious puberty. Adv Pineal Res 1991; 5: 329-331.
- Karasek M, Pawlikowski M, Nowakowska-Jankiewicz B, et al. Circadian variations in plasma melatonin, FSH, LH, prolactin and testosterone levels in infertile men. J Pineal Res 1990; 9: 149-157.
- Puig-Domingo M, Webb SM, Serrano J, et al. Melatonin-related hypogonadotropic hypogonadism. N Engl J Med 1992; 327: 1356-1359.
- Luboshitzky R, Lavi S, Thuma I, Lavie P. Increased nocturnal melatonin secretion in male patients with hypogonadotropic hypogonadism and delayed puberty. J Clin Endocrinol Metab 1995; 80: 2144-2148.
- Luboshitzky R, Wagner O, Lavi S, Here P, Lavie P. Abnormal melatonin secretion in hypogonadal men: the effect of testosterone treatment. Clin Endocrinol 1997; 47: 463-469.
- Berga SL, Mortola JF, Yen SSC. Amplification of nocturnal melatonin secretion in women with functional hypothalamic amenorrhea. J Clin Endocrinol Metab 1988; 66: 242-244.
- Laughlin GA, Loucks AB, Yen SSC. Marked augmentation of nocturnal melatonin secretion in amenorrheic athletes, but not in cycling athletes: Unaltered by opioidergic or dopaminergic blockade. J Clin Endocrinol Metab 1991; 73: 1321-1326.
- Luboshitzky R, Qupti G, Ishay A, Shen-Orr Z, Futrman B, Linn S. Increased 6-sulfatoxymelatonin excretion in women with polycystic ovary syndrome. Fertil Steril 2001; 76: 506-510.
- Brown GM. Pineal function in psychiatric disorders. Front Horm Res 1996; 21: 174-179.
- Wetterberg L. Melatonin and clinical application. Reprod Nutr Dev 1999; 39: 367-382.
- Pacchierotti C, Iapichino S, Bossini L, Pieraccini F, Castrogiovanni P. Melatonin in psychiatric disorders. Front Neuroendocrinol 2001; 22: 18-32.
- Wetterberg L, Aperia B, Gorelick DA, et al. Age, alcoholism and depression are associated with low levels of urinary melatonin. J Psychiatry Neurosci 1992; 17: 215-224.
- Srinivasan V, Pandi-Perumal SR, Maestroni MJG, Esquifino A, Harderland R, Cardinali DP. Role of melatonin in neurodegenerative diseases. Neurotoxicity Res 2005; 7: 293-318.
- Wu YH, Swaab DF. The human pineal gland and melatonin in aging and Alzheimers disease. J Pineal Res 2005; 38: 145-152.
- Fahn S, Cohen G. The oxidant stress hypothesis in Parkinsons disease: evidence supporting it. Ann Neurobiol 1991; 32: 804-812.
- Miller JW, Selhub J, Joseph JA. Oxidative damage caused by free radicals produced during catecholamine autooxidation: protective effects of O-methylation and melatonin. Free Radical Biol Med 1996; 21: 241-249.
- Karasek M, Pawlikowski M. Pineal gland, melatonin and cancer. Neuroendocrinol Lett 1999; 20: 139-144.
- Pawlikowski M, Winczyk K, Karasek M. Oncostatic action of melatonin: facts and question marks. Neuroendocrinol Lett 2002; 23(suppl. 1): 24-29.
- Bartsch C, Bartsch H, Karasek M. Melatonin in clinical oncology. Neuroendocrinol Lett 2002; 23(suppl 1): 30-38.
- Lissoni, P. Efficacy of melatonin in the immunotherapy of cancer using interleukin-2. In The Pineal Gland and Cancer, C Bartsch, H Bartsch, DE Blask, DP Cardinali, WJM Hrushesky, D Mecke (eds). Berlin, Springer, 2001, pp. 465-475.
- Hrushesky, WJM. Melatonin cancer therapy. In The Pineal Gland and Cancer, C Bartsch, H Bartsch, DE Blask, DP Cardinali, WJM Hrushesky, D Mecke (eds). Berlin, Springer, 2001, pp. p. 476-508.
- Karasek M, Reiter RJ. Melatonin and aging. Neuroendocrinol Lett 2002; 23 (suppl. 1): 14-16.
- Kunz D, Mahlberg R, Muller C, Tilmann A, Bes F. Melatonin in patients with reduced REM sleep duration: two randomized controlled trials. J Clin Endocrinol Metab 2004; 89: 128-134.
- Karasek M, Reiter RJ, Cardinali DP, Pawlikowski M. The future of melatonin as a therapeutic agent. Neuroendocrinol Lett 2002; 23 (suppl 1): 118-121.