Obesity is one of the strongest risk factor for the development of OSAS. Obesity is present in 60 to 90% of patients diagnosed with apnea (8). OSAS is three times frequenting among obese than non-obese patients (9). Obesity is associated with metabolic derangement, including dyslipidemia, hyperinsulinism, impaired glucose tolerance and hypertension. The presence of these factors is termed the insulin-resistant syndrome and they predispose obese patients to morbidities such as diabetes mellitus and cardiovascular diseases (10). Recent studies support a correlation between breathing disturbances during sleep and insulin resistant syndrome, independent of the degree of obesity (10-12). The research into the genetic basis of obesity may uncover factors linking obesity and OSAS. The adipose associated hormone - leptin, seems to be responsible for the central regulation of food intake and energy hemostasis, may play a particularly important protective role in the regulation of respiration, especially during sleep (13). The circulating levels of leptin are higher in women than in men because subcutaneous fat (lower - female body obesity) produces more leptin than visceral (upper -male body obesity) (14).
The aim of our study was to evaluate the correlation between the Body Mass Index (BMI) and sleep study parameters in overweight and obese patients suffering from breathing disturbances during sleep.
A total of 106 consecutive obese or overweight patients, represented by 12 women and 94 men, aged from 24 to 78 were recruited for our study. All patients gave their consent to participate in the study, which had previously been approved by the Ethics Committee of the Silesian Medical University. All subjects were presented to our outpatient clinic between July 1997 and April 2002 with a primary complaint of snoring or other breathing disturbances during sleep. All of them were studied during sleep by PolyMESAM (MAP Medizintechnik für artz und patient GMBH). The following parameters were measured: the snoring level, nasal and mouth airflow, body position, heart rate frequency, thoracic and abdominal respiratory movements, oxygen saturation and leg movement activity. On the base of the results after 7-hour night examination the following indexes were calculated: Respiratory Disturbance index (RDI), Apnea Index (AI), Desaturation Index (DI) and Average of Lowest Saturation (LSAT). After the sleep studies our patients were divided into the habitual snorers (RDI<10) and OSA patients (RDI>10). All the patients were physically examined (especially status and anatomical abnormalities of the upper airway). The site of obstruction was identified by means of endoscopic evaluation. On the basis of the BMI values (assessed from patient's weight and height) the subjects were divided into three groups according to the severity of obesity. The first group consisted of subjects with BMI between 25 and 30 kg/m2 (overweight), the second with BMI between 30 and 35 kg/m2 and the third with BMI over the 35 kg/m2.
In these three groups correlations between BMI and sleep study parameters (RDI, AI, DI and LSAT) were examined. The measurements were conducted using the rank correlation Spearman test. The evaluations were performed with the use of the statistical package SPSS and SAS (WIN).
The mean age of the 106 subjects (represented by 12 women and 94 men) was 49.2 ± 11.8 (ranged from 24 to 78 years). This group was divided into three subgroups - overweight (BMI 25-30), obesity class I (BMI 30-35) and obesity class II (BMI over 35). In the first group there were 55 subjects aged from 24 to 71 (mean age 50.4 ± 11). The mean BMI in this group was 27.4 ± 1.4 kg/m2 (ranged from 25 to 29.9). Overnight sleep study revealed that in this group were 22 habitual snorers (RDI lower than 10) and 33 OSA patients (RDI higher than 10). The mean value of RDI was 19.8 ± 20.3 (ranged from 0 to 83). Among the OSA patients, 17 were classified as mild, 10 as moderate and 6 as severe OSAS (Table 1).
| Table 1. The results of sleep study in the overweight and obese patients. |
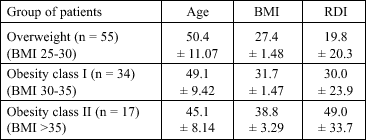 |
The group of class I obesity consisted of 34 patients with BMI between 30 and 35 kg/m2 aged from 27 to 70 years (mean age 49.1 ± 9.4). The mean BMI was 31.7 ± 1.47 kg/m2 (ranged from 30 to 34.9). Sleep study divided obesity subjects into 9 habitual snorers and 25 OSA patients. The mean value of RDI in OSA patients was 30. There were 5 mild, 11 moderate and 9 severe cases of OSAS.
The group of class II obesity consisted of 17 patients with BMI over 35 kg/m2. The mean age of this group was 45.1 ± 8.1 (ranged from 36 to 63). The mean BMI was 38.8 ± 3.2 kg/m2 (ranged from 35 to 45.3). After the sleep study these patients were divided on 4 habitual snorers, 4 moderate and 9 severe OSAS. The mean RDI in this group was 49.
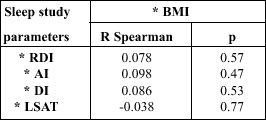
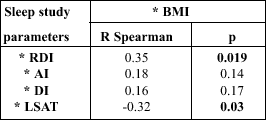
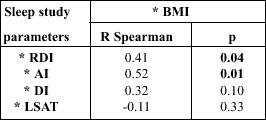
The measurements of relationship between the BMI and sleep study parameters were conducted using the Rang Spearman correlation test. The results of this test were showed in the Table 2, 3 and 4. We found no statistically significant correlation between BMI and sleep study parameters in the overweight patients. In the obesity class I this correlation were statistically significant positive when we compared BMI and RDI and significant negative in comparison of BMI and LSAT. In the obesity class II we found statistically significant positive correlation when we compared BMI with RDI and AI.
| Table 2. The results of correlation test between BMI and sleep study parameters in overweight patients |
 |
| * BMI - Body
Mass Index RDI - Respiratory Disturbance Index AI - Apnea Index DI - Desaturation Index LSAT - Lowest Saturation |
| Table 3. The results of correlation test between BMI and sleep study parameters in obese class I patients (BMI 30-35) |
 |
| * BMI - Body
Mass Index RDI - Respiratory Disturbance Index AI - Apnea Index DI - Desaturation Index LSAT - Lowest Saturation |
| Table 4. The results of correlation test between BMI and sleep study parameters in obese class II patients (BMI over 35) |
 |
| * BMI - Body
Mass Index RDI - Respiratory Disturbance Index AI - Apnea Index DI - Desaturation Index LSAT - Lowest Saturation |
Obesity is probably the most important among the risk factors for OSAS (15, 16). Significant sleep apnea is present in approximately 40% of obese individuals and 70% of OSAS patients are obese. Weight loss in OSAS patients led to a significant decrease in apnea frequency (17-19). An increase in BMI of 1 SD is associated with a fourfold increase in risk for OSA. The neck circumference was the most powerful predictor of OSA among all anthropometric variables. This fact suggested that upper body or central obesity may be important for the development of OSAS (3, 20).
The exact mechanisms underlying the effects of obesity on the risk of OSA are still unclear. It may be the results of fat deposition on upper airway wall or changes in upper airway function (19). The modification in central mechanisms regulating airway tone or ventilatory control stability caused by obesity may also be implicated. Leptin is a circulating hormone expressed, synthesized and released in adipose tissue. Plasma leptin concentrations are increased in people with obesity in direct proportion to body fat mass (3). Leptin has important effect on regulation of chemoreflex function and hence breathing control (19).
It has been recognized that not only increased body weight, but also the type of regional fat distribution (abdominal-visceral vs. gluteal-femoral) plays an important role in the development of OSAS. Central (visceral) obesity is associated more often with OSAS than other forms of obesity. The fat deposition in the neck region, especially in the parapharyngeal region plays an important role in the development of OSAS (3). Fat deposits in the upper airway and pharyngeal walls of obese patients can lead to narrowing of the pharyngeal space (9). Fat infiltration of the upper airway and parapharyngeal region may soften the soft palate, tongue, epiglottis and pharyngeal walls, predisposing to upper airway narrowing or obstruction during sleep (21).
Several studies of obese patients have shown that the relationship between the measures of obesity and severity of OSAS is only moderate and varies widely. Increased BMI has been found to be an unfavourable prognostic factor for the success of surgical treatment for OSAS (22). Akita et al. observed no significant correlation between BMI and either AHI or desaturation rate in obese patients. There was only a tendency for the increase of AHI together with the increase in BMI (23). Vgontzas found in a group of 250 obese men and women that only 50% of men and 8.5% of women had AHI more than 30 during the night. There was no difference in mean BMI in the group with and without apnea (24). Similarly, Rajala found no difference in mean BMI in a group of morbidly obese patients with and without apnea during sleep (25). Fogel observed no relationship between obesity (weight or BMI) and apnea severity (RDI) in the group of 14 morbidly obese patients (4). Dixon studied the predicting factors of OSA in the severely obese patients. He found that four clinical and two biochemical factors independently predicted AHI: observed sleep apnea, male sex, higher BMI, age, fasting insulin and glycosylated hemoglobin A. The increasing of BMI was associated with reduced sleep efficiency and a reduction in the proportion of REM sleep (26). Pillar observed in the large population of patients referred to sleep laboratory the significant correlation between BMI and AHI. This correlation was moderate and weakened when the group was restricted to cases with only overweight (27).
Our results were very similar. We observed no significant relationship between the obesity measurement (BMI) and sleep study parameters (RDI, AI, HI and DI) in overweight patients (BMI 25-30 kg/m2) but in obese patients (class I and II) there were a significant correlation between increasing BMI and RDI was found. The mean RDI progressively increased with the increasing of obesity measurement (BMI).
We conclude that BMI is a simple, yet important predictor of the OSAS in the group of obese patients.
- Antczak J, Geisler P, Popp R. Outcomes of CPAP treatment in a sleep laboratory specialized in neuropsychiatry. J Physiol Pharmacol 2004; 55, supp 3: 15-22.
- Paglietti MG. Sleep disordered breathing in children. J Physiol Pharmacol 2004; 55, supp 3: 164-165.
- Schafer H, Pauleit D, Sudhop T, Gouni-Berthold I, Ewig S, Berthold HK. Body fat distribution, serum leptin, and cardiovascular risk factors in men with obstructive sleep apnea. Chest 2002; 122: 829-839.
- Fogel RB, Malhotra A., Dalagiorgou G, et al. Anatomic and physiologic predictors of apnea severity in morbidly obese subjects. Sleep 2003; 26: 150-155.
- Carden KA, Fogel RB. Obesity-associated hypoventilation - a "growing" concern. Am J Med 2004; 116: 58-59.
- Yu X, Fujimoto K, Urushibata K, Matsuzawa Y, Kubo K. Cephalometric analysis in obese and nonobese patients with obstructive sleep apnea syndrome. Chest 2003; 124: 212-218.
- Tafil-Klawe M, Klawe JJ. Role of nose breathing in genioglossus muscle response to hypoxia in older and younger subjects. J Physiol Pharmacol 2003; 54, supp 1: 48-55.
- Kyzer S, Charuzi I. Obstructive sleep apnea in obese. World J Surg 1998; 22: 998-1001.
- Nishimura Y, Nishimura T, Hattori H, Hattori C, Yonekura A, Suzuki K. Obesity and obstructive sleep apnea syndrome. Acta Otolaryngol Suppl 2003; 550: 22-24.
- Ng DKK, Lam YY, Kwok KL, Chow PY. Obstructive sleep apnea syndrome and obesity in children. Hong Kong Med J 2004; 10: 44-48.
- Punjabi NM, Sorkin JD, Katzel LI, Goldberg AP, Schwartz AR, Smith PL. Sleep-disordered breathing and insulin resistance in middle-aged and overweight men. Am J Respir Cri Care Med 2002; 165: 677-682.
- Vgontzas AN, Bixler EO, Chrousos GP. Metabolic disturbances in obesity versus sleep apnoea: the importance of visceral obesity and insulin resistance. J Intern Med 2003; 254: 32-44.
- Jaworek J, Bonior J, Konturek SJ, Bilski J, Szlachcic A, Pawlik WW. Role of leptin in the control of postprandial pancreatic enzyme secretion. J Physiol Pharmacol 2003; 54, 4: 591-602.
- O'Donnell ChP, Schwartz AP, Smith PL. Upper airway collapsibility. Am J Respir Crit Care Med 2000; 162: 1606-1607.
- Herer B, Roche N, Carton M, Roig C, Poujol V, Huchon G. Value of clinical, functional, and oximetric data for the prediction of obstructive sleep apnea in obese patients. Chest 1999; 116: 1537-1544.
- Hanacek J, Olos R, Vysehradsky R, Snahnicanova B. What pathomechanisms are involved in the development of sleep-related breathing disorders in diabetics? J Physiol Pharmacol 2004; 55, supp 3: 160.
- Kansanen M, Vanninen E, Tuunainen A, et al. The effect of a very low-calorie diet-induced weight loss on the severity of obstructive sleep apnoea and autonomic nervous function in obese patients with obstructive sleep apnoea syndrome. Clin Physiol 1998; 18: 377-385.
- Sampol G, Munoz X, Sagales MT, et al. Long-term efficacy of dietary weight loss in sleep apnoea/hypopnoea syndrome. Eur Respir J 1998; 12: 1156-1159.
- Wolk R, Shamsuzzaman AS, Somers VK. Obesity, sleep apnea, and hypertension. Hypertension 2003; 42(6): 1067-1074.
- Mortimore IL, Marshall I, Wraith PK, Sellar RJ, Douglas NJ. Neck and total body fat deposition in nonobese and obese patients with sleep apnea compared with that in control subjects. Am J Respir Crit Care Med 1998; 157: 280-283.
- Liao YF, Chuang ML, Huang CS, Tsai YY. Upper airway and its surrounding structures in obese and nonobese patients with sleep-disordered breathing. Laryngoscope 2004; 114: 1052-1059.
- Rollheim J, Osnes T, Miljeteig H. The relationship between obstructive sleep apnoea and body mass index. Clin Otolaryngol 1997; 22: 419-422.
- Akita Y, Kawakatsu K, Hattori Ch, Hattori H, Suzuki K, Nishimura T. Posture of patients with sleep apnea during sleep. Acta Otolaryngol 2003; 550: 41-45.
- Vgontzas AN, Tan TL, Bixler EO, Martin LF, Shubert D, Kales A. Sleep apnea and sleep disruption in obese patients. Arch Intern Med 1994; 154: 1705-1711.
- Rajala R, Partinen M, Sane T, Pelkonen R, Huikuri K, Seppalainen AM. Obstructive sleep apnea syndrome in morbidly obese patients. J Intern Med 1991; 230: 125-129.
- Dixon JB, Schachter LM, O'Brien PE. Predicting sleep apnea and excessive day sleepiness in the severely obese: indicators for polysomnography. Chest 2003; 123: 1134-1141.
- Pillar G, Peled N, Katz N, Lavie P. Predictive value of specific risk factors, symptoms and signs, in diagnosing obstructive sleep apnea and its severity. J Sleep Res 1994; 3: 241-244.